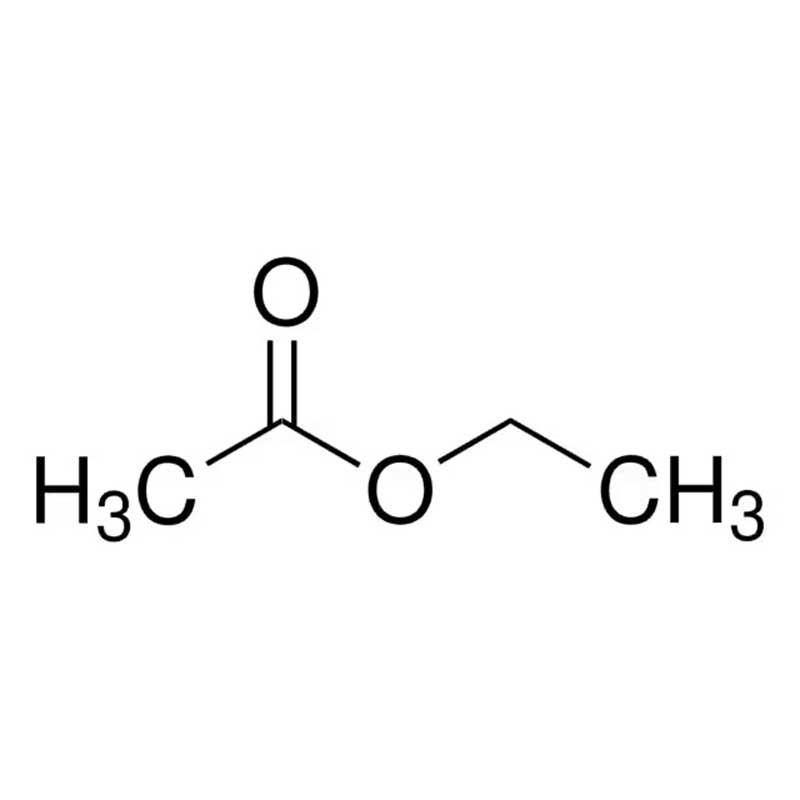
Ethyl Acetate CAS : 141-78-6
Ethyl acetate (CH₃COOC₂H₅) is a colorless, volatile liquid with a characteristic fruity aroma. It is an important organic solvent with strong dissolving power, widely used in the production of paints, inks, and adhesives. It also serves as a common fruit-flavored food flavoring in the food industry and is a key raw material in pharmaceuticals and fragrance synthesis. Known for its low toxicity, fast evaporation, and pleasant odor, ethyl acetate finds extensive applications in both industrial and consumer products.
I. Basic Characteristics Overview
Ethyl acetate, with the chemical formula CH₃COOCH₂CH₃, is an organic compound formed by the esterification of acetic acid and ethanol. It is a colorless, transparent, highly volatile liquid with a strong, pleasant fruity odor (reminiscent of pineapple or pear). It possesses excellent solvent properties, capable of dissolving a wide range of organic substances, making it one of the most widely used organic solvents. It is indispensable in industries such as coatings, inks, adhesives, pharmaceuticals, and food. Ethyl acetate is highly flammable, and its vapors can form explosive mixtures with air, necessitating strict attention to fire and explosion prevention during its production, storage, and use.
II. Key Physical & Chemical Properties
| Property | Value / Description |
|---|---|
| Chemical Name | Ethyl Acetate |
| Chemical Formula | C₄H₈O₂ / CH₃COOC₂H₅ |
| Molecular Weight | 88.11 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Sweet, fruity, similar to pineapple or pear |
| Boiling Point | 77.1 °C |
| Melting Point | -83.6 °C |
| Density (20°C) | 0.902 g/mL |
| Flash Point | -4 °C (closed cup) (Highly Flammable Liquid) |
| Autoignition Temperature | 426 °C |
| Explosive Limits (in air) | 2.0% - 11.5% (by volume) |
| Refractive Index (20°C) | 1.3720 |
| Viscosity (25°C) | 0.45 mPa·s |
| Solubility | Slightly soluble in water (~8.7 g/100 mL at 20°C); miscible with most organic solvents like ethanol, ether, acetone, chloroform. |
| Vapor Pressure (20°C) | 97 mmHg |
| Key Chemical Properties | Typical ester; undergoes hydrolysis in the presence of acid or base catalysts to form acetic acid and ethanol; reacts with amines via aminolysis. |
III. Primary Application Fields
Solvent (Primary Use): One of the most common and excellent solvents for nitrocellulose coatings (e.g., nail polish, furniture lacquer), inks, adhesives (e.g., contact cement), and artificial leather production, due to its strong solvency, moderate evaporation rate, and low residue.
Food Industry: Used as a flavoring agent (nature-identical) in fruit-flavored essences for imparting fruity notes to candies, beverages, ice cream, etc.
Pharmaceutical Industry: Serves as an extraction solvent and reaction medium for isolating active ingredients from natural sources (e.g., caffeine from tea) and in drug synthesis processes.
Chemical Intermediate: Used in the synthesis of acetic anhydride, ethyl acetoacetate, pharmaceutical intermediates, and other fine chemicals.
Other Uses: Employed as a cleaning agent for degreasing, as a mobile phase in chromatography, and as a fragrance in cosmetics.
IV. Storage & Handling
Storage Conditions: Must be stored in a cool, well-ventilated dedicated warehouse or explosion-proof cabinet, away from heat sources, ignition sources, and strong oxidizers (e.g., nitric acid, potassium permanganate). The storage temperature should not exceed 30°C. Keep containers tightly sealed.
Packaging Materials: Suitable containers include stainless steel tanks, galvanized steel drums, or epoxy-lined steel drums. Avoid prolonged use of plastic drums (risk of swelling).
Handling Precautions:
Personnel must wear chemical safety goggles, solvent-resistant gloves (e.g., nitrile or Viton), and anti-static work clothing. Appropriate respiratory protection is required in poorly ventilated areas.
Smoking is strictly prohibited in operational areas, and all potential open flames, sparks (including static electricity) must be eliminated. All equipment should be properly grounded, and explosion-proof electrical equipment must be used.
Prevent vapor leaks into the workplace air; ensure effective local exhaust ventilation.
Spill Response: Immediately eliminate ignition sources and evacuate personnel upwind. For small spills, absorb with sand or other inert material. For large spills, construct containment dikes. Cover with alcohol-resistant foam to reduce vapor hazard. Transport collected material to a waste disposal site. Rinse the spill area thoroughly with plenty of water.
Firefighting Measures: Highly flammable liquid. Preferred extinguishing agents: alcohol-resistant foam, dry chemical powder, or carbon dioxide. Use water or water mist with caution, as they may spread the fire. Firefighters must wear self-contained breathing apparatus and full protective clothing. Cool exposed containers with water spray.