Acetic Acid CAS: 64-19-7
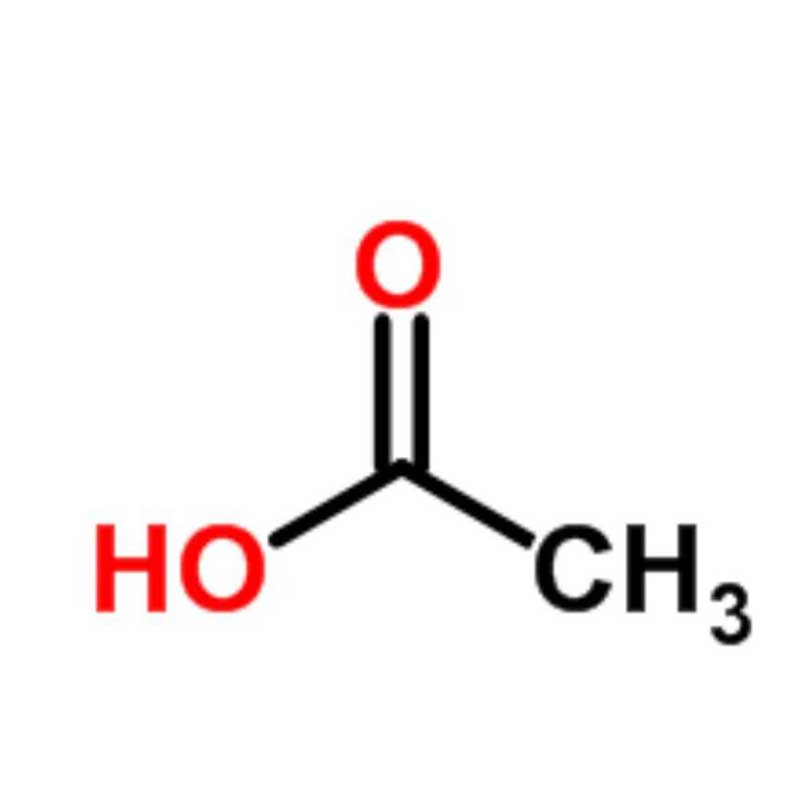
Acetic acid (CH₃COOH) is the main component of vinegar and a vital organic chemical raw material. In its pure form, it is a colorless liquid with a pungent odor, which solidifies into ice-like crystals at low temperatures (hence called glacial acetic acid). It is widely used as an acidity regulator in the food industry and is a key intermediate in the production of synthetic fibers, pharmaceuticals, dyes, and agrochemicals.
I. Basic Characteristics Overview
Acetic acid, with the chemical formula CH₃COOH, is one of the simplest members of the carboxylic acid family (second only to formic acid). It is both a crucial organic chemical raw material and a common substance in daily life (edible vinegar contains approximately 3-9% acetic acid). The pure substance (glacial acetic acid) is a colorless, transparent crystal at room temperature (below 16.6°C) and a colorless liquid with a pungent, sour odor at ambient temperatures. Acetic acid exhibits the typical chemical properties of carboxylic acids, such as acidity and esterification. It is readily soluble in water, ethanol, ether, and other solvents. It is widely used in numerous industrial fields including synthetic fibers, pharmaceuticals, food, and dyeing, making it a fundamental bulk chemical.
II. Key Physical & Chemical Properties
| Property | Value / Description |
|---|---|
| Chemical Name | Acetic Acid, Ethanoic Acid |
| Chemical Formula | CH₃COOH / C₂H₄O₂ |
| Molecular Weight | 60.05 g/mol |
| Appearance (Ambient) | Colorless transparent liquid |
| Odor | Pungent, sour odor (vinegar-like) |
| Freezing Point | 16.6 °C (pure substance, hence "glacial" acetic acid) |
| Boiling Point | 118.1 °C |
| Density (20°C) | 1.049 g/mL |
| Flash Point | 39 °C (closed cup) |
| Autoignition Temperature | 427 °C |
| Refractive Index (20°C) | 1.3718 |
| Viscosity (25°C) | 1.22 mPa·s |
| Solubility | Miscible with water, ethanol, glycerol, ether; insoluble in carbon disulfide. |
| Vapor Pressure (20°C) | 15.7 mmHg |
| pKa (25°C) | 4.76 (weak acid) |
| Key Chemical Properties | Exhibits general carboxylic acid properties: acidity (reacts with bases, metal oxides, active metals), undergoes esterification to form acetate esters, and can be converted to derivatives like acyl chlorides and anhydrides. |
III. Primary Application Fields
Chemical Raw Material (Primary Use): The foundational raw material for producing numerous key chemicals including Vinyl Acetate Monomer (VAM) (for polyvinyl acetate, polyvinyl alcohol, and vinylon fiber), Purified Terephthalic Acid (PTA) (for polyester fiber and PET plastic), Acetate Esters (e.g., ethyl acetate, butyl acetate, used as solvents), Acetic Anhydride, and Chloroacetic Acid.
Food Industry: Used as an acidity regulator, preservative, and flavoring agent (food-grade acetic acid) in vinegar, condiments, pickled foods, salad dressings, etc.
Pharmaceutical Industry: An important intermediate and reaction solvent for synthesizing various drugs, including Aspirin (acetylsalicylic acid) and Paracetamol/Acetaminophen.
Solvent: Itself and its derived esters are widely used as organic solvents in the paint, coating, ink, and adhesive industries.
Textile Dyeing: Used as a neutralizing agent, pH adjuster, and co-solvent for certain dyes, also in the manufacture of cellulose acetate.
Other Uses: Used in producing synthetic detergents, bactericides, photographic chemicals, and as a de-icing agent (potassium acetate solution), among others.
IV. Storage & Handling
Storage Conditions: Must be stored in a cool, dry, and well-ventilated dedicated warehouse, away from ignition sources, heat, and oxidizers (e.g., nitrates, permanganates). The storage temperature is preferably maintained above 16°C (to prevent solidification; if solidified, thaw slowly using a warm water bath). Containers must be tightly sealed, preferably under a nitrogen blanket to prevent moisture absorption.
Packaging Materials: Suitable containers include aluminum vessels, stainless steel tanks, glass bottles, or polyethylene plastic drums. Copper, brass, zinc, and similar metal containers are strictly prohibited due to corrosion.
Handling Precautions:
Personnel must wear chemical safety goggles or a face shield, acid-resistant gloves (e.g., butyl rubber), protective clothing resistant to permeation, and respiratory protection (especially in poorly ventilated areas).
Ensure effective local exhaust ventilation due to its strong corrosiveness and irritancy.
Smoking, heat, and open flames are strictly prohibited in the operational area.
Use explosion-proof tools and equipment during handling or transfer operations.
Spill Response: For small spills, mix with dry sand, soda ash, or inert absorbent material and collect in designated containers. For large spills, dike and contain, then neutralize with alkaline substances (e.g., sodium bicarbonate, calcium hydroxide). Finally, flush thoroughly with copious amounts of water, treating the wastewater to meet standards before discharge.
Firefighting Measures: Flammable liquid. Use water spray, alcohol-resistant foam, dry chemical powder, or carbon dioxide for firefighting. Firefighters must wear self-contained breathing apparatus and full protective clothing. Cool exposed containers with water spray.