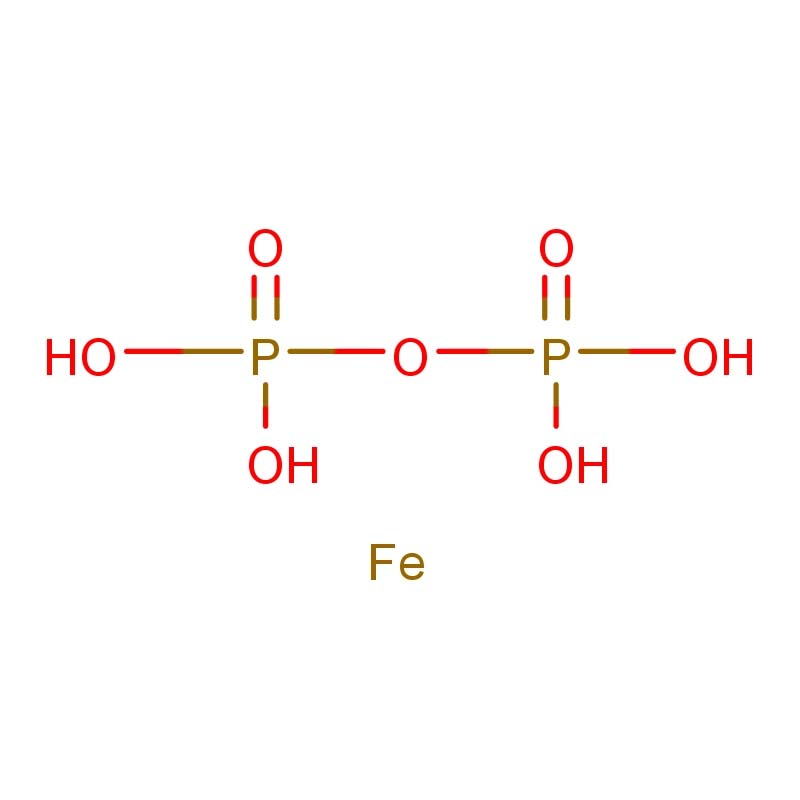
Ferric pyrophosphate
Excellent Acid and Alkali Solubility: Demonstrates ready solubility in inorganic acids, alkaline solutions, and citric acid, facilitating its use in food fortification and nutritional supplements.
Sensory Neutrality: Effectively releases iron in the gastric environment without altering the taste or appearance of food products, thereby ensuring high consumer acceptability.
High Hygroscopicity and Stability: Exhibits moisture-absorbing properties while remaining stable under standard ambient conditions, making it suitable for diverse industrial applications.
Controlled Release Profile: Its insolubility in cold water enables targeted iron release primarily in acidic conditions, allowing for precise bioavailability.
1. Product Overview
Ferric pyrophosphate is an inorganic iron compound widely recognized as a safe, stable, and effective source of iron for food fortification and pharmaceutical applications. Its chemical formula is typically represented as Fe₄(P₂O₇)₃, with CAS number 10058-44-3 and molecular weight of approximately 745.22 g/mol (anhydrous basis) or 925.38 g/mol for the common decahydrate Fe₄(P₂O₇)₃·10H₂O .
This compound appears as a yellowish-white to tan or pale yellow powder, odorless and stable under normal storage conditions . Its defining characteristic is the ability to provide bioavailable iron while remaining sensorially neutral—it does not cause the unacceptable color changes, off-flavors, or metallic taste typically associated with more soluble iron salts like ferrous sulfate .
Ferric pyrophosphate exists in two primary forms:
Standard (insoluble) ferric pyrophosphate: Used in food fortification where minimal sensory impact is required
Soluble ferric pyrophosphate complexes: Modified forms (e.g., ferric pyrophosphate citrate, FPC) developed for pharmaceutical applications and enhanced bioavailability
The compound's unique position in the iron supplement landscape stems from balancing efficacy with tolerability—it provides needed iron while minimizing the gastrointestinal side effects that often lead to poor compliance with traditional iron therapy .
2. Key Features
Superior Tolerability Profile: Demonstrates significantly fewer gastrointestinal side effects compared to conventional ferrous sulfate. Clinical studies show GI adverse events in only 12% of pediatric patients receiving ferric pyrophosphate versus 41% with ferrous sulfate (p < 0.01) . In pregnant women, emulsified microsomal ferric pyrophosphate showed zero adverse effects versus 11.1% with ferrous ascorbate .
Sensorially Neutral: Unlike water-soluble iron salts, standard ferric pyrophosphate does not cause unacceptable color changes, off-flavors, or rancidity in fortified foods, making it ideal for food fortification applications .
Enhanced Bioavailability via Nanotechnology: Nano-sizing and advanced formulations (liposomal, emulsified, micronized) dramatically improve iron absorption. Coated ferric pyrophosphate nanoparticles (FPP-NPs) achieve in-vitro bioaccessibility of 63.78%, comparable to ferrous sulfate, compared to only 19.86% for unfortified controls .
Dual Mechanism of Absorption: Absorbed via enterocytes and specialized M cells through both paracellular and transcellular routes, often as intact particles. This unique absorption pathway contributes to improved gastrointestinal tolerance .
Multiple Advanced Formulations Available:
Emulsified Microsomal Ferric Pyrophosphate (EMFP) : SunActive™ Fe, showing efficacy at 1/3 the dose of conventional iron
Liposomal Ferric Pyrophosphate: Demonstrates superior hematological outcomes and better compliance in children
Sucrosomial® Iron: Ferric pyrophosphate conveyed by phospholipid and sucrester matrix
Nanoparticle formulations: For enhanced bioavailability with minimal sensory impact
Established Safety Profile: FDA-approved for food fortification and supplement formulation . Acute and chronic toxicity studies in animal models demonstrate favorable safety with established LD₅₀ values .
Comparable Efficacy at Lower Doses: Achieves equivalent hemoglobin improvement at significantly lower elemental iron doses. Pregnant women receiving EMFP (27 mg iron) showed similar Hb increase to those receiving ferrous ascorbate (100 mg iron) .
3. Technical Specifications with Explanations
| Parameter | Typical Value/Specification | Description & Significance |
|---|---|---|
| Molecular Formula | Fe₄(P₂O₇)₃ (anhydrous) Fe₄(P₂O₇)₃·10H₂O (decahydrate) | Core identifier; exists in hydrated and anhydrous forms . |
| Molecular Weight | 745.22 g/mol (anhydrous) 925.38 g/mol (decahydrate) | Used for stoichiometric calculations and formulation . |
| CAS Number | 10058-44-3 | Universal chemical identifier for ferric pyrophosphate . |
| Appearance | Yellowish-white to tan or pale yellow powder | Visual quality indicator; color may vary slightly by hydration state and manufacturing process . |
| Odor | Odorless | Sensory characteristic critical for food applications . |
| Iron Content | ~24-26% (anhydrous basis) ~10.5-12.5% (in soluble complexes) | Elemental iron content varies by form and hydration . |
| Solubility in Water | Insoluble (standard form) Highly soluble (>1000 mg/mL for FPC) | Critical differentiator. Standard form insoluble → minimal sensory impact; soluble complexes for pharmaceutical use . |
| Solubility in Acids | Slightly soluble in mineral acids | Dissolves in gastric environment for absorption . |
| pH (suspension) | Near-neutral | Minimal impact on food matrices . |
| Stability | Stable in air; decomposes on heating | Heat-stable up to ~300°C (nanoparticle formulations) . |
| Particle Size | Variable by grade: • Standard: microns to mm range • Nanoparticles: ~121 nm • Micronized: 0.3-0.77 mm | Smaller particles dramatically increase surface area and bioavailability . |
| Zeta Potential | ~ ±30 mV (nanoparticle formulations) | Indicates colloidal stability of nanoparticle suspensions . |
| Bioaccessibility | ~64% (coated FPP-NPs) ~54% (uncoated FPP-NPs) ~20% (unfortified controls) | Measures iron available for absorption; improved by nanotechnology . |
Soluble Ferric Pyrophosphate Citrate (FPC) Specifications
| Parameter | Typical Value | Significance |
|---|---|---|
| Composition | Fe(III) complexed with citrate and pyrophosphate | Enhanced solubility and stability |
| Molecular Formula | Complex; e.g., C₆H₅Fe₂NaO₁₄P₂ | Variable based on stoichiometry |
| Appearance | Yellow to green amorphous powder | Visual quality indicator |
| Solubility | >1000 mg/mL in water | Enables parenteral and high-concentration applications |
| Iron Content | ~10.5-12.5% | Suitable for therapeutic use |
| Solution Stability | Stable for several months | Practical for pharmaceutical manufacturing |
4. Applications
Food Fortification & Nutrition
Staple Food Fortification: Added to wheat flour, rice, and other staples to combat iron deficiency without affecting taste or color . Standard insoluble ferric pyrophosphate is preferred when sensory neutrality is paramount.
Dairy Product Fortification:
Paneer (Indian cottage cheese) : Fortified with coated FPP nanoparticles at up to 25% incorporation maintains acceptable sensory characteristics (hedonic rating 8.20/10) .
Milk powders and yogurt: Provides iron fortification without lipid oxidation or off-flavors.
Beverage Fortification: Soluble ferric pyrophosphate complexes (e.g., SunActive™ Fe) enable clear beverage fortification without precipitation or color changes .
Infant Formulas and Cereals: Iron-fortified complementary foods for infants and young children, using micronized forms to enhance bioavailability .
Nutritional Supplements: Tablet, capsule, and powder formulations for over-the-counter and prescription iron supplements.
Pharmaceutical & Clinical Applications
Oral Iron Therapy:
Pregnancy: Emulsified microsomal ferric pyrophosphate (27 mg Fe) as effective as ferrous ascorbate (100 mg Fe) with superior tolerability in second-trimester pregnant women .
Pediatrics: Effective in children aged 6 months to 12 years at 1 mg/kg/day (vs. 3 mg/kg/day for ferrous sulfate) with significantly fewer GI side effects .
Liposomal Formulations: Liposomal SunActive™ iron demonstrates better hematological outcomes, reduced drug refusal rates, and improved compliance in children .
Parenteral Nutrition: Soluble ferric pyrophosphate citrate (FPC) developed for intravenous administration, particularly valuable for patients with chronic kidney disease undergoing hemodialysis .
Iron Deficiency Intolerant to Conventional Salts: First-line alternative for patients who cannot tolerate ferrous sulfate due to gastrointestinal side effects .
Advanced Drug Delivery Systems
Nanoparticle Formulations:
Coated with ascorbic acid and folic acid, stabilized with whey protein concentrate for enhanced absorption .
Surface modification with dextrin and glycerides (SunActive™) to improve dispersibility and intestinal transport .
Sucrosomial® Technology: Ferric pyrophosphate conveyed by phospholipid and sucrester matrix (sucrosome®) enabling absorption as intact particles with excellent gastrointestinal tolerance .
Research Applications
Bioavailability Studies: Used in Caco-2 cell models and in vivo research to understand iron metabolism and absorption mechanisms .
Nanotechnology Research: Model compound for developing novel iron delivery systems with enhanced bioavailability and minimal sensory impact .
Toxicity Assessments: Reference compound for acute and chronic oral toxicity studies, with established LD₅₀ and histopathological profiles .
5. Product Grade Comparison
| Parameter/Grade | Food Grade (Standard) | Micronized Food Grade | Pharmaceutical Grade (FPC) | Nanoparticle Formulations | Liposomal/Sucrosomial® |
|---|---|---|---|---|---|
| Core Standard | Food additive specifications | Enhanced bioavailability grade | Meets pharmacopoeia standards | Proprietary nanotechnology | Proprietary delivery systems |
| Typical Purity | ≥99% | ≥99% | ≥98% | ≥95% (as iron equivalent) | ≥95% (as iron equivalent) |
| Iron Content | ~24-26% | ~24-26% | ~10.5-12.5% | Variable (10-26%) | Variable (10-20%) |
| Particle Size | 0.5-5 mm | 0.3-77 μm | Molecular dispersion | ~121 nm | Micron to submicron |
| Solubility | Insoluble in water | Insoluble | Highly soluble | Dispersible | Dispersible |
| Bioavailability | Low-moderate | Moderate (62-82% RBA) | High | Very high (comparable to FeSO₄) | Very high |
| GI Tolerance | Excellent | Excellent | Good | Excellent | Excellent |
| Sensory Impact | Minimal | Minimal | N/A (pharmaceutical) | Minimal | N/A |
| Primary Applications | Flour, rice, staple foods | Infant cereals, nutritional supplements | Parenteral nutrition, clinical IV use | Advanced food fortification, supplements | Pharmaceutical, clinical nutrition |
| Representative Products | Standard ferric pyrophosphate | SunActive™ Fe (micronized) | Ferric pyrophosphate citrate | Coated FPP nanoparticles | Liposomal SunActive™, Sucrosomial® Iron |
| Regulatory Status | FDA food grade | GRAS | Investigational/approved | Research stage | Commercial pharmaceutical |
| Typical Packaging | 25 kg bags/drums | 20 kg bags | Custom pharmaceutical packaging | Research quantities | Clinical trial supplies |
6. Purchasing & Selection Guide
Select by Application Area
Food & Beverage Manufacturers:
Standard Food Grade: For dry applications (flour, rice, dry mixes) where minimal sensory impact is required and solubility is not needed .
Micronized Grade: For infant cereals, powdered beverages, and applications where enhanced bioavailability is desired without sensory changes .
Soluble/Coated Grades (SunActive™) : For liquid beverages, dairy products, and applications requiring clarity and no sedimentation .
Pharmaceutical & Clinical Manufacturers:
Soluble Ferric Pyrophosphate Citrate (FPC) : For parenteral formulations and intravenous iron therapy .
Liposomal/Sucrosomial® Technology: For oral pharmaceutical formulations targeting patients intolerant to conventional iron salts .
Research Laboratories:
High Purity Grade: For bioavailability studies, cell culture work, and in vivo research
Nanoparticle Formulations: For advanced delivery system development and mechanistic studies
Critical Selection Considerations
Form vs. Function:
Insoluble forms = Best sensory neutrality, lower bioavailability
Micronized forms = Balanced bioavailability and sensory performance
Soluble/complexed forms = Maximum bioavailability, may have sensory impact
Particle Size Drives Performance: Smaller particles dramatically increase surface area and gastric solubility, directly correlating with bioavailability. Relative bioavailability improves from 62% at 0.77 mm to 82% at 0.3 mm .
Regulatory Status Varies by Form:
Standard ferric pyrophosphate is FDA-approved for food fortification
Novel formulations (liposomal, nanoparticle) may require specific regulatory approvals per market
Pharmaceutical forms must meet applicable pharmacopoeia standards
Cost Considerations:
Standard grade: Most economical
Micronized/enhanced forms: Moderate premium
Proprietary technologies (SunActive™, Sucrosomial®): Premium pricing justified by clinical benefits
7. Frequently Asked Questions (FAQ)
Q: What is the difference between standard ferric pyrophosphate and soluble ferric pyrophosphate?
A: Standard ferric pyrophosphate (Fe₄(P₂O₇)₃) is insoluble in water, making it ideal for food fortification where minimal sensory impact is required . Soluble ferric pyrophosphate (e.g., ferric pyrophosphate citrate, FPC) is a complex where iron is chelated with citrate and pyrophosphate, achieving high water solubility (>1000 mg/mL) suitable for pharmaceutical and parenteral applications .
Q: Is ferric pyrophosphate as effective as ferrous sulfate for treating iron deficiency?
A: Yes, with appropriate formulation. Clinical studies demonstrate:
In pregnancy: EMFP (27 mg Fe) equivalent to ferrous ascorbate (100 mg Fe)
In children: FPP (1 mg/kg/day) equivalent to ferrous sulfate (3 mg/kg/day)
Nanoparticle formulations achieve bioaccessibility comparable to ferrous sulfate
The efficacy depends on particle size, formulation technology, and dosing strategy.
Q: Why is ferric pyrophosphate better tolerated than ferrous sulfate?
A: Two key reasons:
Absorption mechanism: Ferric pyrophosphate is absorbed via specialized pathways (enterocytes, M cells, often as intact particles) rather than releasing free iron in the gut lumen .
Controlled iron release: The complex structure prevents the sudden, high local iron concentrations that cause oxidative stress and mucosal irritation. Clinical studies show significantly lower GI side effects: 12% vs. 41% in children ; 0% vs. 11.1% in pregnant women .
Q: Can ferric pyrophosphate be used in clear beverages?
A: Standard ferric pyrophosphate is insoluble and will precipitate, making it unsuitable for clear beverages. However, soluble/complexed forms (SunActive™ Fe, ferric pyrophosphate citrate) are designed for clear beverage applications and remain stable without sedimentation or color changes .
Q: How does nanotechnology improve ferric pyrophosphate performance?
A: Nanotechnology (particle size reduction to ~121 nm) provides multiple benefits:
Increased surface area → faster dissolution in gastric juice
Enhanced bioavailability → coated FPP nanoparticles achieve 63.78% bioaccessibility vs. 19.86% for unfortified controls
Improved dispersibility in food matrices
Maintained sensory neutrality while approaching ferrous sulfate efficacy
Potential for targeted delivery with surface coatings
Q: What is SunActive™ Fe and how is it different?
A: SunActive™ Fe (Taiyo Kagaku) is a proprietary emulsified microsomal ferric pyrophosphate where iron is coated with dextrin, glycerides, and lecithin . This technology:
Prevents iron interactions with food components
Enables fortification of challenging matrices (beverages, dairy)
Shows efficacy at 1/3 the dose of conventional iron
Demonstrates zero adverse effects in clinical studies
Enhances cellular uptake and intestinal transport
Q: Is ferric pyrophosphate safe for infants and children?
A: Yes, multiple clinical studies support its safety in pediatric populations:
Infants (6-12 months): Used for prophylaxis
Children (6 months-12 years): Effective at 1 mg/kg/day with superior tolerability
Children (2-12 years): Liposomal formulations show better compliance and growth outcomes
Always use age-appropriate formulations and follow pediatric dosing guidelines.
Q: How should ferric pyrophosphate be stored?
A: Standard ferric pyrophosphate is stable at room temperature in sealed containers, protected from moisture . Nanoparticle formulations and liposomal products may have specific storage requirements (cool, dry conditions; protect from extreme temperatures). Always refer to the Certificate of Analysis for batch-specific storage recommendations.
Q: Does ferric pyrophosphate cause dark stools like other iron supplements?
A: Dark stools are a common effect of oral iron therapy due to unabsorbed iron in the gut. While ferric pyrophosphate's improved absorption may theoretically reduce this effect, patients should still expect some stool darkening. The 2025 pregnancy study reported dark stools only in the ferrous ascorbate group, suggesting potentially less effect with advanced formulations .
Q: What is the difference between liposomal iron and Sucrosomial® iron?
A: Both are advanced delivery technologies for ferric pyrophosphate:
Liposomal iron: Iron encapsulated within phospholipid bilayers (liposomes); absorbed via M cells and enterocytes
Sucrosomial® iron: Ferric pyrophosphate conveyed by a phospholipid and sucrester matrix (sucrosome®); absorbed as intact particles
Both demonstrate enhanced bioavailability and excellent gastrointestinal tolerance compared to conventional iron salts.
8. Delivery, Certification & Service
Delivery Capabilities
Inventory Status: Multiple grades available from regional warehouses (Asia, Europe, Americas)
Sample Program: Small quantities available for research institutions and manufacturers for formulation testing and evaluation
Packaging Options:
Research quantities: 1 kg, 5 kg containers
Industrial quantities: 25 kg bags/drums (standard)
Bulk quantities: 200 kg drums, 500 kg supersacks, 20 MT containers
Custom packaging: Available for volume customers
Shipping: Not regulated as hazardous for most transport modes; standard freight shipping available
Quality Certifications
Certificate of Analysis (COA) provided with each batch, documenting:
Appearance and color
Purity (≥99% typical)
Iron content
Heavy metals (Pb, As) within limits
Particle size distribution (where applicable)
Safety Data Sheet (SDS) available in multiple languages
Product Specification (PS) documenting chemical composition and physical properties
Certificates of Origin (COO) available upon request
Additional certifications available:
Food-grade compliance documentation
Kosher/Halal certifications (by request)
ISO 9001 quality management certification
GMP documentation for pharmaceutical grades
Regulatory Compliance
Food Grade: Meets international food additive specifications
Novel Foods: Liposomal and nanoparticle formulations may require novel food approvals in some jurisdictions
Pharmaceutical: Compliance with applicable pharmacopoeia standards (USP, Ph.Eur.) for clinical grades
Export Documentation: Full compliance package for international shipping
Technical Support & Services
Technical Consultation: Application specialists available for:
Formulation development and optimization
Bioavailability enhancement strategies
Sensory impact mitigation
Regulatory pathway guidance
Custom Services: Tailored particle size, coating options, and packaging for qualified customers
Clinical Research Support: Documentation and reference materials for clinical trial applications
Documentation Available
Certificate of Analysis (COA) with batch-specific data
Safety Data Sheet (SDS/MSDS)
Technical Data Sheet (TDS)
Product Specification (PS)
Certificate of Origin (COO)
Food-grade compliance statements
Kosher/Halal certificates (where applicable)
GMP documentation (pharmaceutical grades)
Stability data (by request)