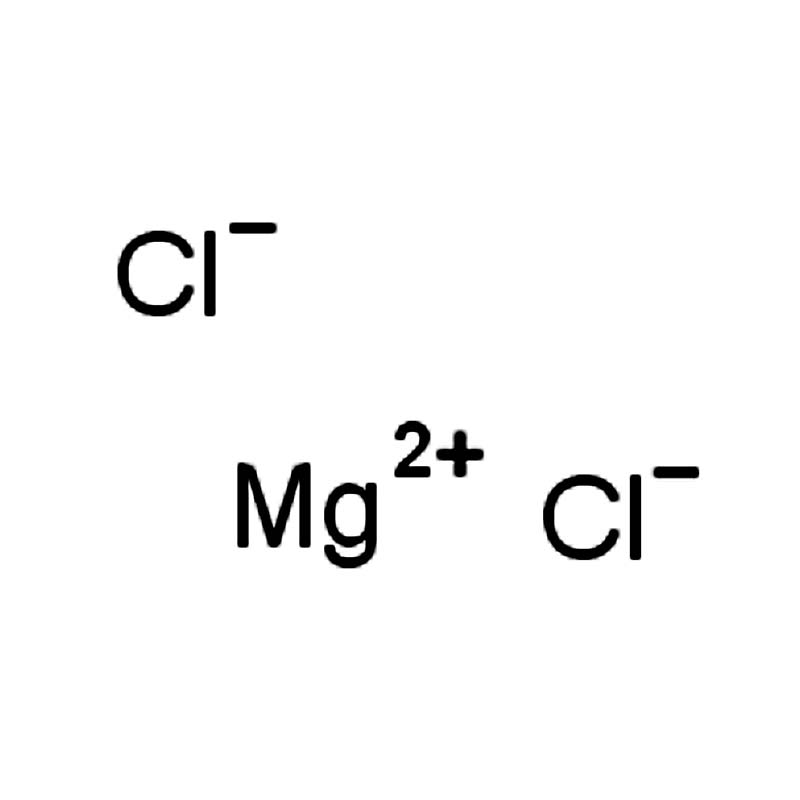
Magnesium dichloride CAS:7786-30-3
Anhydrous magnesium chloride is produced through dehydration or specific processes (such as thermal decomposition of magnesium ammonium chloride compound salts, or molten electrolysis of magnesium chloride, etc.), removing the crystalline water and some impurities (such as sulfate, boron, etc.) that were carried along with the water.
This makes it a key raw material for the production of high-purity magnesium metal (through molten salt electrolysis). Impurities can seriously affect the efficiency of the electrolysis and the purity of the magnesium.
High water absorption: Magnesium chloride without water is an efficient desiccant and moisture absorber, suitable for applications requiring deep drying.
As a catalyst or precursor: In organic chemistry (such as the Ziegler-Natta catalyst), the absence of water is essential because water would poison the catalyst.
Preparation of other high-purity magnesium compounds: This is an ideal starting point for the production of high-performance materials such as high-purity magnesium oxide and magnesium hydroxide.
Anhydrous magnesium chloride (Magnesium dichloride) is a biochemical reagent that can be used as a biological material or an organic compound in life science-related research.
2. Physical and chemical properties of magnesium chloride:
| Density | 2.32 g/mL at 25 °C(lit.) |
| Boiling point | 1412°C |
| Melting point | 714 °C(lit.) |
| Molecular formula | Cl2Mg |
| Molecular weight | 95.21 |
| Precise quality | 93.922745 |
| InChIKey | TWRXJAOTZQYOKJ-UHFFFAOYSA-L |
| SMILES | [Cl-].[Cl-].[Mg+2] |
| Appearance and properties | White powder |
| Refractive index | n20/D 1.336 |
| Storage conditions | 2-8℃ |
| Stability | If used and stored in accordance with the specifications, it will not decompose. |
| Avoid contact with acids, moisture/dampness. Its hygroscopicity is stronger than that of other alkaline earth metal chlorides. Heating to 300°C releases chlorine gas and forms chlorides. | |
| Water solubility | 400 G/L (20 ºC) |
3: Composition/information on ingredients
| Chemical name | Common names and synonyms | CAS number | EC number | Concentration |
| Magnesium chloride | Magnesium chloride | 7786-30-3 | 232-094-6 | 100% |
4: First-aid measures
Description of necessary first-aid measures
If inhaled
Fresh air, rest.
Following skin contact
Rinse skin with plenty of water or shower.
Following eye contact
Rinse with plenty of water for several minutes (remove contact lenses if easily possible).
Following ingestion
Rinse mouth.
Most important symptoms/effects, acute and delayed
no data available
Indication of immediate medical attention and special treatment needed, if necessary
Absorption, Distribution and Excretion
Three mature ewes each received by intrarumenal infusion a supplement of 0, 1, 2 and 3 g Mg/d in sequence over four 10-day periods. Net Mg absorption distal to the pylorus took place down its electrochemical gradient, although the quantity absorbed remained small during the control and first infusion periods. The bulk of Mg absorption occurred before the pylorus and, during the control and first infusion periods, took place against its electrochemical gradient. The net Mg absorption proximal to the pylorus rose with declining efficiency as Mg intake was increased. It is suggested that saturation of the absorption process at this site was occurring.
5: Accidental release measures
Personal precautions, protective equipment and emergency procedures
Personal protection: particulate filter respirator adapted to the airborne concentration of the substance. Sweep spilled substance into covered containers. If appropriate, moisten first to prevent dusting.
Environmental precautions
Personal protection: particulate filter respirator adapted to the airborne concentration of the substance. Sweep spilled substance into covered containers. If appropriate, moisten first to prevent dusting.
Methods and materials for containment and cleaning up
Collect and arrange disposal. Keep the chemical in suitable and closed containers for disposal. Remove all sources of ignition. Use spark-proof tools and explosion-proof equipment. Adhered or collected material should be promptly disposed of, in accordance with appropriate laws and regulations.
6: Handling and storage
Precautions for safe handling
Handling in a well ventilated place. Wear suitable protective clothing. Avoid contact with skin and eyes. Avoid formation of dust and aerosols. Use non-sparking tools. Prevent fire caused by electrostatic discharge steam.
Conditions for safe storage, including any incompatibilities
Dry.
7: Transport information
UN Number
ADR/RID: Not dangerous goods. (For reference only, please check.) IMDG: Not dangerous goods. (For reference only, please check.) IATA: Not dangerous goods. (For reference only, please check.)
UN Proper Shipping Name
ADR/RID: Not dangerous goods. (For reference only, please check.) IMDG: Not dangerous goods. (For reference only, please check.) IATA: Not dangerous goods. (For reference only, please check.)
Transport hazard class(es)
ADR/RID: Not dangerous goods. (For reference only, please check.) IMDG: Not dangerous goods. (For reference only, please check.) IATA: Not dangerous goods. (For reference only, please check.)
Packing group, if applicable
ADR/RID: Not dangerous goods. (For reference only, please check.) IMDG: Not dangerous goods. (For reference only, please check.) IATA: Not dangerous goods. (For reference only, please check.)
Environmental hazards
ADR/RID: No IMDG: No IATA: No
Special precautions for user
no data available
Transport in bulk according to IMO instruments
no data available