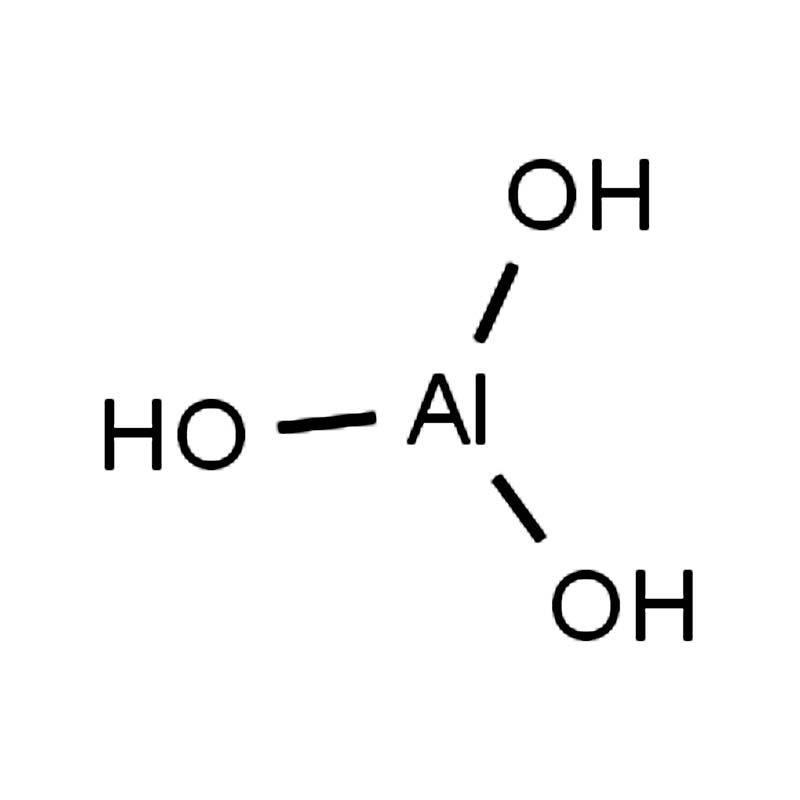
Aluminium hydroxide CAS:21645-51-2
Aluminum hydroxide is a widely used chemical product. Industrial-grade aluminum hydroxide is mainly used as a filler for plastics and polymers, a flame retardant and adhesive for weaving, a filler for epoxy resins, a color filler for papermaking and coatings, for the production of aluminum sulfate, alum, aluminum fluoride, sodium aluminate, for the synthesis of molecular sieves, as a filler for toothpaste, a component of glass, its gel solution and Chemicalbook's dried gel. Pharmaceutical-grade aluminum hydroxide is mainly used as an antacid, having the effect of neutralizing stomach acid and protecting ulcer surfaces, which can alleviate symptoms such as excessive stomach acid combined with acid reflux. It is suitable for the treatment of gastric ulcers and duodenal ulcers, reflux esophagitis, and upper gastrointestinal bleeding. When used in combination with calcium supplements and vitamin D, it can treat neonatal hypocalcemia (tetany).
Aluminum hydroxide (Al(OH)3) is a white solid, insoluble in water but soluble in acids and alkalis, and is a typical amphoteric hydroxide. Upon heating in air, it dehydrates and transforms into aluminum oxide, which is of great importance in alumina production. Aluminum hydroxide reacts with both acids and strong alkalis to form chemical salts and water, thus it is an amphoteric hydroxide. Because it also exhibits some acidity, it can be called aluminic acid (H3AlO3). However, when it actually reacts with alkalis, it forms tetrahydroxyaluminate ([Al(OH)4]-). Therefore, it is usually considered as monohydrate aluminic acid (HAlO2·H2O), and is classified into industrial and pharmaceutical grades according to its uses.
| Melting point | 300℃ |
| Boiling point | 2980C[at 101 325 Pa] |
| Bulk density | ~90g/100 mL |
| Density | 2.42 g/cm3 at 20 °℃ |
| Vapor pressure | <0.1 hPa (20 °C) |
| Refractive index | Average refractive index: 1.57-1.59 |
| Storage conditions | Store at +5°C to +30°℃. |
| Solubility | The solubility in water is 0.0015 grams per liter. |
| Form | Colloidal suspension |
| Color | white |
| Specific gravity | 2.42 |
| Odor | Flavorless |
| pH value | 8-9 (100g/l, H2O, 20°℃)(slurry) |
| The pH range at which acid-base indicators change color | >7 |
| Water solubility | insoluble |
| Crystal structure | Monoclinic |
| Merck | 14,342 |
| Ksp - Solubility Product Constant | pKsp: 32.89 |
| Exposure limit | ACGIH: TWA 1 mg/m3 |
| Dielectric constant | 2.2 (Ambient) |
| Stability | Stable,Not compatible with strong bases. |
| Main application | pharmaceutical (small molecule) |
| Cosmetic ingredient efficacy | SKIN CONDITIONING-EMOLLIENT OPACIFYING HUMECTANT SKIN PROTECTING VISCOSITY CONTROLLING COLORANT |
| Cosmetic ingredient assessment | Aluminum hydroxide (21645-51-2) |
| InChl | 1S/Al.3H2O/h;3*1H2/q+3;/p-3 |
| InChlKey | WNROFYMDJYEPJX-UHFFFAOYSA-K |
| SMILES | O[AI](O)O |
| LogP | -1.380 (est) |
| CAS Database | 21645-51-2(CAS DataBase Reference) |
| EPA Chemical Substance Information | Aluminum hydroxide (21645-51-2) |
Application
1. Chemical raw materials
Because aluminum hydroxide can be produced on a large scale, with sufficient raw materials, high product purity, and easy solubility in acids and bases. Therefore, aluminum hydroxide is an important raw material for preparing aluminum salts, such as barium aluminate and aluminum sulfate.
2. Flame retardant
Aluminum hydroxide powder, due to its functions of filling, flame retardancy, and smoke suppression, and being non-toxic and harmless, is usually regarded as an ideal flame retardant filler for plastics, unsaturated polyesters, rubber, and other organic polymers. The flame retardation mechanism of aluminum hydroxide: When the temperature exceeds 200℃, aluminum hydroxide begins to absorb heat and decompose and release three crystalline water. At around 250℃, its decomposition rate is the highest. That is: This reaction is a strong endothermic reaction, thereby inhibiting the increase in polymer temperature and reducing its decomposition rate, and only generating water vapor, without generating toxic and harmful gases.
3. Ceramics
Aluminum hydroxide is obtained by high-temperature calcination of aluminum oxide, and aluminum oxide has high thermal chemical stability, high strength, anti-rheological property, dielectric property, and low thermal expansion coefficient, and is an important material for synthesizing ceramics. In the ceramic synthesis process, aluminum hydroxide activates and controls the crystallization process, thereby achieving the control of phase formation in composite materials.
4. Sewage treatment
Aluminum hydroxide in water exists mainly as Al(OH)4-, which can precipitate toxic heavy metals in the sewage through the co-precipitation method and then filter to achieve the effect of purifying water. Aluminum hydroxide also has a high specific surface area, and can adsorb colloids, suspended solids, dyes, organic substances, etc. on its surface.
5. Medicine
Aluminum hydroxide can neutralize stomach acid and is non-toxic and harmless, and is a traditional good medicine for treating stomach diseases. And using aluminum hydroxide as an adjuvant can improve the immunogenicity of vaccines. Its mechanism of action is to adsorb antigens on the surface of aluminum hydroxide, allowing them to release slowly, achieving the effect of prolonging the drug efficacy.
6. Catalyst carrier
Due to the fact that during the preparation process of aluminum hydroxide, different products with different specific surface areas, pore volumes, pore structures, and crystal structures can be obtained by controlling reaction conditions such as reaction temperature, concentration, and pH, it can effectively be used as a catalyst carrier for the hydrogenation of unsaturated carbonyl compounds and the preparation of fullerene, etc.
7. Paper industry
Aluminum hydroxide has high whiteness, ultra-fine particle size, complete crystal form, and strong compatibility with whitening agents. When used as an additive for coatings and resins, it can effectively improve the whiteness, opacity, smoothness, and inkability of coated paper.