I. Basic Characteristics Overview
Diethylene glycol (DEG), also known as diglycol, is a chemical compound with the formula C₄H₁₀O₃. It is a primary byproduct in the production of ethylene glycol. It appears as a colorless, odorless (or with a faint, sweet odor), transparent, highly hygroscopic, viscous liquid. Its chemical properties are similar to those of ethylene glycol, exhibiting the typical reactivity of alcohol hydroxyl groups, such as esterification and etherification. Diethylene glycol is highly toxic and strictly prohibited for use in food, pharmaceuticals, and cosmetics. Its primary applications are concentrated in industrial sectors, where it serves as a solvent, plasticizer, and chemical intermediate.
II. Key Physical & Chemical Properties
| Property | Value / Description |
|---|---|
| Chemical Name | Diethylene Glycol, Diglycol |
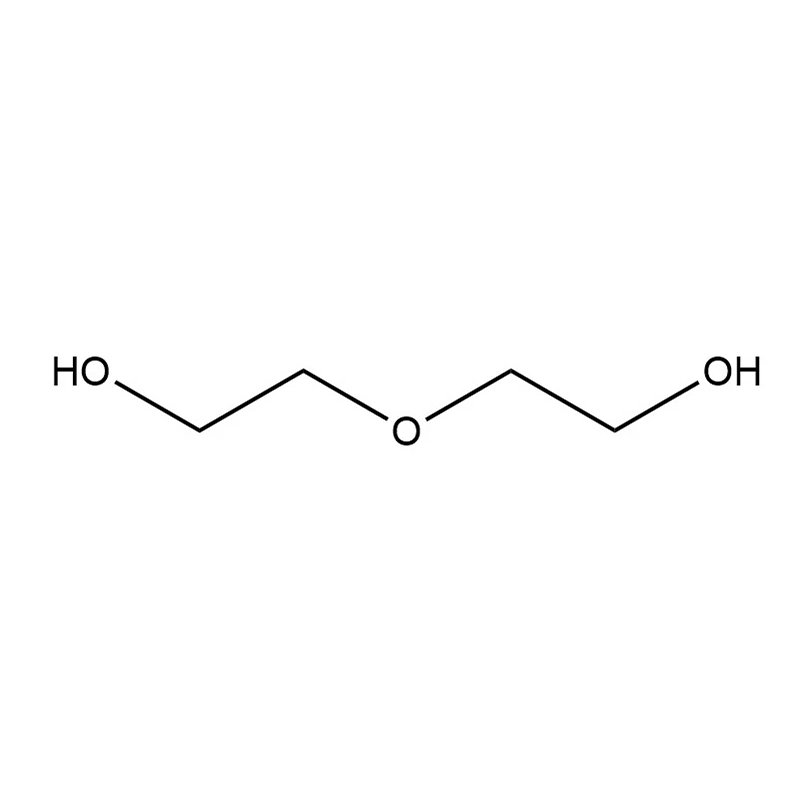
| Chemical Formula | C₄H₁₀O₃ or HO-CH₂-CH₂-O-CH₂-CH₂-OH |
| Molecular Weight | 106.12 g/mol |
| Appearance | Colorless, transparent, viscous liquid |
| Odor | Odorless or faintly sweet alcoholic odor |
| Density (20°C) | ~1.118 g/mL |
| Boiling Point | 245 °C (at atmospheric pressure) |
| Melting/Freezing Point | -10.5 °C |
| Flash Point | 143 °C (closed cup) |
| Autoignition Temperature | ~229 °C |
| Refractive Index (20°C) | ~1.447 |
| Viscosity (20°C) | ~35 mPa·s |
| Solubility | Miscible with water, ethanol, acetone, ether; insoluble in non-polar solvents like benzene, toluene. |
| Vapor Pressure (20°C) | Very low (< 1 Pa) |
| Key Chemical Properties | Contains two primary alcohol hydroxyl groups; can undergo esterification, etherification, oxidation; highly hygroscopic. |
III. Primary Application Fields
Gas Dehydration & Desiccant: Due to its strong hygroscopicity, it is a common dehydrating agent and desiccant for industrial gases like natural gas and refinery gas.
Plasticizer & Solvent: Used as a solvent for nitrocellulose, resins, and oils, and as a reactive plasticizer or solvent in industries such as polyurethane and unsaturated polyester.
Chemical Intermediate: An important raw material for producing chemicals like morpholine and 1,4-dioxane; also used to synthesize solvent series like diethylene glycol ethers (carbitols).
Textiles & Lubricants: Used as a lubricant and softening agent in textile dyeing and printing; can formulate specialty lubricants and brake fluids.
Other Industrial Uses: A component in adhesives and printing inks, and used as an antifreeze for concrete (industrial use, no human contact).
Critical Warning: Diethylene glycol possesses nephrotoxicity and neurotoxicity. Historically, its accidental use in pharmaceuticals (e.g., the Elixir Sulfanilamide disaster) and toothpaste (e.g., the Panama deaths) has caused mass poisoning fatalities. It is absolutely forbidden for use in any consumer product that may involve human ingestion or prolonged contact.
IV. Storage & Handling
Storage Conditions: Store in a cool, dry, well-ventilated area, away from ignition sources, heat, and strong oxidizers (e.g., peroxides, nitric acid). Keep containers tightly sealed to prevent moisture absorption.
Packaging Materials: Suitable containers include stainless steel, galvanized steel drums, or polyethylene plastic drums. Avoid prolonged use of aluminum containers.
Handling Precautions:
Personnel must wear chemical safety goggles, chemical-resistant gloves (e.g., nitrile rubber), and protective clothing is recommended.
Ensure adequate ventilation in the workplace, especially in confined spaces. Avoid inhalation of vapors or mists.
Due to its toxicity, strictly prohibit eating, drinking, or smoking in the work area. Wash thoroughly after handling.
Spill Response: Contain and absorb with sand or inert absorbent material. Collect into designated containers. Flush contaminated area with large amounts of water. Direct wastewater to treatment systems.
Firefighting Measures: Combustible liquid. Use water fog, foam, dry chemical powder, or carbon dioxide for firefighting. Firefighters must wear self-contained breathing apparatus and full protective clothing.