Lithium Carbonate
High Stability: Lithium carbonate maintains excellent stability under atmospheric conditions without hygroscopicity, ensuring long-term storage integrity and straightforward handling.
Controlled Solubility Profile: It exhibits limited water solubility, characterized by an inverse solubility profile where dissolution decreases with rising temperature, suitable for applications requiring minimal aqueous interaction.
Thermal Resilience: Possessing a melting point of 618°C, the compound demonstrates exceptional thermal endurance for high-temperature industrial processes.
Broad Functional Utility: It serves critical roles in lithium-ion battery cathode production, ceramic glaze formulations, and as a psychotropic medication for bipolar disorder management.
1. Product Overview
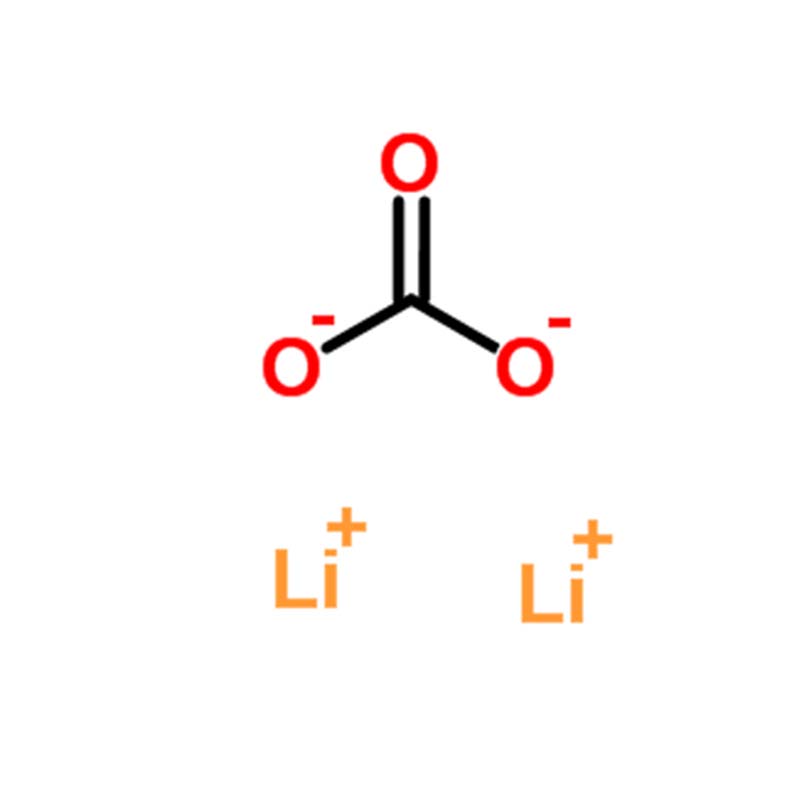
Lithium carbonate (Li₂CO₃) is the most important industrial compound of lithium, serving as the precursor for virtually all other lithium compounds. It is an inorganic salt that appears as a white, odorless crystalline powder or colorless monoclinic crystal . Its chemical formula is Li₂CO₃ with CAS number 554-13-2 and molecular weight of approximately 73.89 g/mol .
Lithium carbonate occupies a unique position across multiple industries—from its critical role in lithium-ion batteries powering the electric vehicle revolution to its established use as a first-line mood stabilizer in psychiatry. It is also widely used in glass, ceramics, aluminum production, and specialty applications .
First isolated in the early 19th century, lithium carbonate's psychiatric applications were discovered in the 1940s, while its importance in energy storage has skyrocketed in the 21st century with the global transition to electrification.
2. Key Features
Dual Market Criticality: Serves as the foundational material for both the energy storage industry (battery-grade) and the pharmaceutical industry (USP-grade), with purity specifications tailored to each application .
Battery-Grade Purity: High-purity lithium carbonate (≥99.5%) with extremely low levels of impurities (Fe, Cu, Zn, Pb < 0.0003%) is essential for synthesizing cathode materials like lithium iron phosphate (LFP), lithium cobalt oxide, and ternary materials .
Pharmaceutical-Grade Efficacy: USP-grade lithium carbonate is a first-line treatment for bipolar disorder, effectively managing manic episodes and providing maintenance therapy to prevent recurrence . It is available in both immediate-release and extended-release formulations .
Thermal Stability: With a melting point of 618-735°C and boiling point of 1310-1342°C (decomposition), it remains stable across a wide temperature range, making it suitable for high-temperature industrial processes .
Controlled Solubility: Slightly soluble in water (1.29 g/100 mL at 25°C) but readily soluble in dilute acids, allowing controlled release in pharmaceutical applications and controlled reactions in industrial processes .
Critical Material for Green Transition: As the primary raw material for lithium-ion batteries, it plays an essential role in electric vehicles, energy storage systems, and portable electronics, supporting global decarbonization efforts .
Multiple Purity Grades Available: Offered in various specifications from technical grade (99%) through high-purity battery grade (99.5-99.99%) to ultra-high purity (99.999%) for specialized applications .
3. Technical Specifications with Explanations
| Parameter | Typical Value/Specification | Description & Significance |
|---|---|---|
| Molecular Formula | Li₂CO₃ | Core identifier; consists of two lithium cations and one carbonate anion . |
| Molecular Weight | 73.89 g/mol | Used for stoichiometric calculations and formulation . |
| CAS Number | 554-13-2 | Universal chemical substance identifier . |
| EINECS Number | 209-062-5 | European inventory identifier . |
| Appearance | White crystalline powder or colorless monoclinic crystals | Visual quality indicator; should be free from discoloration or visible impurities . |
| Odor | Odorless | Sensory characteristic . |
| Assay (Purity) | 99% – 99.999% (grade dependent) • Technical/Reagent: 99% • Battery grade: ≥99.5% • High purity: 99.99-99.999% | Core specification. Higher purity critical for battery performance and pharmaceutical safety . |
| Melting Point | 618°C – 735°C (varies by source) • 618-723°C • 720°C • 723°C • 735°C | Characteristic physical constant; variations reflect different measurement methods and grades. |
| Boiling Point | 1310°C – 1342°C (decomposes) | Very high thermal stability before decomposition . |
| Density | 2.11 – 2.112 g/cm³ | Slightly denser than water; used for volume-to-weight conversions . |
| Solubility in Water | 1.29 g/100 mL (25°C) Slightly soluble | Limited water solubility enables controlled release in pharmaceuticals and controlled precipitation in industrial processes. |
| Solubility in Acids | Soluble in dilute acids | Readily dissolves in acid, allowing conversion to other lithium compounds . |
| Solubility in Organic Solvents | Insoluble in alcohol and acetone | Remains undissolved in organic media . |
| pH (saturated solution) | ~11 (alkaline) | Basic character due to carbonate hydrolysis. |
| Specific Heat | 97.4 J/mol·K | Thermal property for process engineering . |
| Refractive Index | 1.428 – 1.482 | Optical property for quality control . |
| Hygroscopicity | Poor, hygroscopic | Absorbs moisture from air; requires sealed storage . |
| Stability | Stable under normal conditions; decomposes at high temperatures | Avoid contact with acids (evolves CO₂) and strong oxidizing agents . |
Battery Grade Specifications (Typical)
| Parameter | Specification | Significance |
|---|---|---|
| Li₂CO₃ Purity | ≥ 99.60% | Core quality metric |
| SO₄²⁻ | ≤ 0.080% | Sulfate impurity affects battery performance |
| Na | ≤ 0.020% | Sodium interferes with cathode performance |
| Mg | ≤ 0.0060% | Magnesium affects electrochemical properties |
| Ca | ≤ 0.0040% | Calcium impurity control |
| Fe | ≤ 0.0005% | Iron critical for battery performance |
| Cu | ≤ 0.0003% | Copper control essential |
| Pb | ≤ 0.0003% | Heavy metal limit |
| Magnetic impurities (Fe+Cr+Zn) | ≤ 150 ppb | Critical for battery safety |
| Cl | ≤ 0.002% | Chloride limit |
| H₂O | ≤ 0.25% | Moisture control critical |
| Particle size D50 | 5.0-8.0 µm | Optimal for cathode processing |
Pharmaceutical Considerations
| Parameter | Therapeutic Range | Toxic Level | Significance |
|---|---|---|---|
| Therapeutic serum level | 0.8 – 1.2 mEq/L | N/A | Optimal efficacy with minimal side effects |
| Toxic threshold | N/A | ≥ 1.5 mEq/L | Narrow therapeutic window requires monitoring |
| Half-life | ~24 hours | N/A | Once-daily dosing possible |
| Bioavailability | 95-100% | N/A | Well absorbed orally |
4. Applications
Energy Storage & Battery Industry
Lithium-Ion Battery Cathodes: Primary use of battery-grade lithium carbonate is in synthesizing cathode materials:
Lithium Iron Phosphate (LFP): Fast-growing cathode material for electric vehicles and energy storage
Lithium Cobalt Oxide (LCO): Used in consumer electronics
Lithium Manganese Oxide (LMO) and ternary materials (NCM, NCA): For high-performance EV batteries
Battery-Grade Lithium Compounds: Converted to lithium hydroxide for high-nickel cathodes or other specialty lithium chemicals
Pharmaceutical & Medical Applications
Bipolar Disorder Treatment: First-line maintenance therapy for manic-depressive illness, reducing the frequency and severity of manic episodes
Available as immediate-release and extended-release tablets (150 mg, 300 mg, 400 mg, 450 mg, 600 mg)
Extended-release formulations reduce peak-to-trough variability in blood levels
Mania Management: Effective for acute manic episodes, typically normalizing symptoms within 1-3 weeks
Investigational Uses: Studied for cognitive effects, though evidence does not show significant memory impairment at therapeutic doses
Historical Cardiac Use: Investigated in refractory congestive heart failure for diuretic effects
Glass & Ceramics Industry
Glass Manufacturing: Reduces melting temperature and thermal expansion; improves strength and clarity in specialty glasses, ovenware, and glass-ceramics
Ceramic Glazes: Used in both low-fire and high-fire ceramic glazes as a flux; improves color development and reduces firing time
Tile Adhesives: Enhances bonding properties in construction materials
Aluminum Production
Electrolyte Additive: Added to aluminum smelting baths (Hall-Héroult process) to lower melting point and increase current efficiency
Industrial & Specialty Applications
Lithium Compounds Production: Starting material for manufacturing all other lithium chemicals (lithium hydroxide, lithium chloride, lithium bromide, etc.)
Metal Oxides Processing: Used in processing of various metal oxides for specialty applications
Catalysts: Component in catalyst formulations for chemical synthesis
High-Technology Applications
Optical Grade: For specialty glasses, optical components, and photonics
Electronics: Used in semiconductor manufacturing and electronic components
Single Crystals: For producing lithium tantalate and lithium niobate single crystals used in acoustic wave devices and optical applications
5. Product Grade Comparison
| Parameter/Grade | Technical/Reagent Grade | Battery Grade | Pharmaceutical Grade (USP) | High Purity (99.99-99.999%) |
|---|---|---|---|---|
| Core Standard | General industrial/lab use | Battery manufacturer specifications | Meets USP pharmacopoeia standards | Ultra-high purity for critical applications |
| Purity (Assay) | 99% | ≥99.5-99.9% | Meets USP requirements | 99.99-99.999% |
| Critical Parameters | Assay, solubility, appearance | Extremely low impurities (Fe, Cu, Pb, magnetic particles), particle size control | Heavy metals, purity, dissolution profile | Maximum purity, minimal trace elements |
| Typical Impurity Limits | Less stringent | Fe ≤ 5 ppm, Cu ≤ 3 ppm, Pb ≤ 3 ppm | Meets USP monograph | ppb-level controls |
| Physical Form | White powder | Controlled particle size powder | Powder for compression | High-purity powder |
| Primary Applications | Laboratory use, general industrial, research | Lithium-ion battery cathodes (LFP, NCM, LCO) | Bipolar disorder medication, pharmaceutical compounding | Advanced batteries, optics, electronics, single crystals |
| Packaging | 500 g bottles, 25 kg bags | 20 kg bags, 500 kg supersacks, moisture-proof | Sealed containers with desiccant | Hermetically sealed, inert atmosphere |
| Certifications | Manufacturer COA | COA with full trace element analysis, ROHS report | USP certification, DMF available | Full elemental analysis |
| Storage Requirements | Ambient, dry | ≤50°C, ≤95% RH, sealed, moisture-proof | Controlled room temperature, protect from moisture | Controlled environment, inert gas |
| Shelf Life | 2-5 years | 12 months | As indicated on packaging | Limited, use promptly |
| Typical Price Position | Reference | +50-100% | +100-200% | +300-500% |
6. Purchasing & Selection Guide
Select by Application Area
Battery Manufacturers:
Fe, Cu, Pb each ≤ 0.0003% (3 ppm) maximum
Magnetic impurities ≤ 150 ppb (Fe+Cr+Zn)
Particle size D50 5.0-8.0 µm for optimal cathode processing
Moisture ≤ 0.25% (critical for battery performance)
Must select Battery Grade with ≥99.5% purity and strict impurity controls
Critical parameters requiring verification:
Request COA with full trace element analysis and ROHS report (annual)
Verify consistency across batches (every 5 tons tested)
Consider shelf life: 12 months maximum for battery grade
Pharmaceutical Manufacturers:
Immediate-release vs. extended-release formulations
Tablet strength options (150, 300, 400, 450, 600 mg)
Inactive ingredients compatibility
Drug Master File (DMF) access
cGMP manufacturing certification
USP monograph compliance verification
Must select USP Grade meeting pharmacopoeia standards
Require comprehensive documentation:
Consider formulation requirements:
Understand narrow therapeutic index requiring serum monitoring
Glass & Ceramic Manufacturers:
Technical Grade (99%) typically sufficient
Focus on consistency and impurity profiles that affect color and melting
Verify particle size for uniform mixing
Consider cost-effectiveness for high-volume applications
Research Laboratories:
Reagent Grade (99%) for general research
High Purity (99.99-99.999%) for specialized studies
Analytical standards for quantitative analysis
Chemical Processors:
Select grade based on end-product requirements
Battery-grade starting material for high-purity downstream products
Technical grade for industrial-scale conversions
Critical Selection Considerations
Purity Requirements Drive Selection:
Battery applications demand the strictest impurity controls, especially for magnetic particles
Pharmaceutical applications require toxicological safety and consistent dissolution
Glass/ceramics prioritize cost-effectiveness and consistent melting behavior
Physical Form Matters:
Powder: Standard for most applications
Controlled particle size: Essential for battery cathode manufacturing
Compressible grade: Required for tablet formulation
Moisture Control:
Battery grade: Strict moisture limits (≤0.25%)
Pharmaceutical: Moisture affects tablet stability
All grades: Hygroscopic nature requires sealed packaging
Regulatory & Safety Considerations
⚠️ Important Regulatory Notes:
Pharmaceutical Use: Lithium has a narrow therapeutic index (0.8-1.2 mEq/L); toxic levels begin at ≥1.5 mEq/L
Pregnancy Warning: Lithium may cause fetal harm, including cardiac anomalies (Ebstein's anomaly)
Nursing Mothers: Lithium excreted in breast milk; nursing not recommended
Pediatric Use: Safety in children under 12 not established
TSCA Status: Listed for US commerce
REACH: Registered for EU
Transport: Not regulated as hazardous for most modes
7. Frequently Asked Questions (FAQ)
Q: What is the difference between battery-grade and pharmaceutical-grade lithium carbonate?
A: These grades are optimized for completely different applications:
Battery-grade (≥99.5%) focuses on ultra-low metallic impurities (Fe, Cu, Pb ≤ 3 ppm) and magnetic particle control (≤150 ppb) because these affect battery performance and safety. Particle size is also controlled (5-8 µm) for cathode manufacturing .
Pharmaceutical-grade (USP) prioritizes consistent dissolution profile, bioavailability, and toxicological safety. It must meet USP monograph requirements and be manufactured under cGMP .
They are not interchangeable—using the wrong grade can lead to product failure or safety issues.
Q: Is lithium carbonate safe for treating bipolar disorder?
A: Lithium carbonate is a first-line, FDA-approved treatment for bipolar disorder when used under proper medical supervision. However, it has a narrow therapeutic window (0.8-1.2 mEq/L)—levels above 1.5 mEq/L can be toxic . Regular serum monitoring is essential. Side effects can include tremor, polyuria, thyroid issues, and in rare cases, more serious effects . Always use only under professional medical care.
Q: Why is lithium carbonate critical for electric vehicle batteries?
A: Lithium carbonate is the primary raw material for synthesizing cathode materials used in lithium-ion batteries. For electric vehicles, high-purity lithium carbonate is converted to cathode active materials like lithium iron phosphate (LFP) or precursor for high-nickel ternary materials (NCM, NCA) . The purity directly impacts battery performance, cycle life, and safety—hence the stringent specifications for battery grade.
Q: What are the hazards of handling lithium carbonate?
A: Lithium carbonate is classified with the following hazards:
Harmful if swallowed (H302) — oral rat LD50: 525 mg/kg
Causes serious eye irritation (H319)
May cause irritation to skin and respiratory tract
Not flammable, but may emit toxic fumes when heated to decomposition
Avoid contact with acids (releases CO₂)
Use appropriate PPE: gloves, safety glasses, protective clothing
Q: How should lithium carbonate be stored?
A: Storage requirements vary by grade:
General: Store in tightly sealed containers in cool, dry, well-ventilated area
Battery grade: Temperature ≤50°C, relative humidity ≤95%, moisture-proof packaging, avoid air contact
All grades: Protect from moisture (hygroscopic), keep away from acids and strong oxidizing agents
Shelf life: Battery grade 12 months ; other grades typically 2-5 years under proper conditions
Q: Why are there different melting point values reported?
A: The reported melting point ranges from 618-735°C depending on:
Measurement method and conditions
Purity grade (impurities affect melting)
Crystalline form
Heating rate
All values are within acceptable range for this compound .
Q: Can lithium carbonate be used in glass making?
A: Yes, it is widely used in specialty glass and ceramics as a flux to reduce melting temperature, improve strength, and enhance optical properties. It's particularly valuable in ovenware, glass-ceramics, and technical glasses .
Q: What is the difference between immediate-release and extended-release lithium carbonate?
A:
Immediate-release: Rapid absorption, higher peak levels, typically requires multiple daily doses
Extended-release: Designed to release drug gradually, reducing peak-to-trough variability in blood levels, often enabling once-daily dosing
The choice depends on patient tolerance and treatment goals, determined by prescribing physician.
Q: How is lithium carbonate produced?
A: Commercial production methods include:
From lithium-containing brines: Lithium extracted from salt lake brines, purified, and precipitated with sodium carbonate
From spodumene ore: Ore is roasted, converted to lithium sulfate, then precipitated with sodium carbonate
From recycled batteries: Emerging source as recycling technology advances
High-purity grades require additional purification steps.
8. Delivery, Certification & Service
Delivery Capabilities
Inventory Status: Multiple grades available from regional warehouses (US, Europe, Asia)
Sample Program: Small quantities available for qualification and testing
Packaging Options:
Research quantities: 100g, 500g, 1kg, 5kg bottles or bags
Industrial quantities: 20kg, 25kg paper-plastic composite bags, fiber drums
Bulk quantities: 500kg supersacks, palletized with stretch wrap, 1 ton pallets
Full container loads: 20 MT for large-volume customers
Custom packaging: Available for volume customers (IBC totes, special labeling)
Shipping: Not regulated as hazardous for most transport modes (road, rail, sea, air)
Protection: Moisture-proof packaging essential; consider desiccant for humid environments
Quality Certifications
Certificate of Analysis (COA) provided with each batch, documenting:
Appearance
Assay/purity
Impurity profile (grade-appropriate)
Particle size (where applicable)
Moisture content
Safety Data Sheet (SDS) available in multiple languages
Product Specification (PS) documenting chemical composition and physical properties
Certificates of Origin (COO) available upon request
Additional certifications available:
Battery grade: Full trace element analysis, ROHS report (annual)
Pharmaceutical grade: USP certification, DMF access, cGMP compliance statement
ISO 9001 quality management certification
REACH compliance documentation
TSCA statement
Regulatory Compliance
Pharmaceutical: USP monograph compliance
Battery: Customer-specific specifications based on cathode chemistry
Food/Pharmaceutical excipient: Not applicable (therapeutic ingredient)
Export Documentation: Full compliance package for international shipping
Technical Support & Services
Technical Consultation: Application specialists available for:
Battery-grade specifications and testing
Pharmaceutical formulation support
Industrial process optimization
Custom Services: Tailored particle size, purity specifications, and packaging for qualified customers
Regulatory Support: Documentation for product registration and compliance
Documentation Available
Certificate of Analysis (COA) with batch-specific data
Safety Data Sheet (SDS/MSDS)
Technical Data Sheet (TDS)
Product Specification (PS)
Certificate of Origin (COO)
Battery-grade full elemental analysis
USP certification (pharmaceutical grade)
ROHS report (battery grade, annual)
GMP compliance statement (pharmaceutical)
REACH/TSCA statements
Allergen Declaration (none)
GMO-Free Statement (not applicable)