Ethylenediaminetetraacetic acid
Strong Chelating Ability: EDTA exhibits hexadentate chelation, forming highly stable coordination compounds through six-point binding with metal ions.
Effective Ligand: It functions as a potent polydentate ligand, enabling widespread use in industrial and laboratory settings for metal ion complexation.
Wide Application Range: This compound finds utility across diverse sectors including water treatment, pharmaceutical manufacturing, agricultural formulations, and as a specialty additive in chemical processes.
Chemical Stability: EDTA maintains structural integrity at elevated temperatures (up to 250°C) and demonstrates resistance to decomposition, ensuring reliability in demanding applications.
1. Product Overview
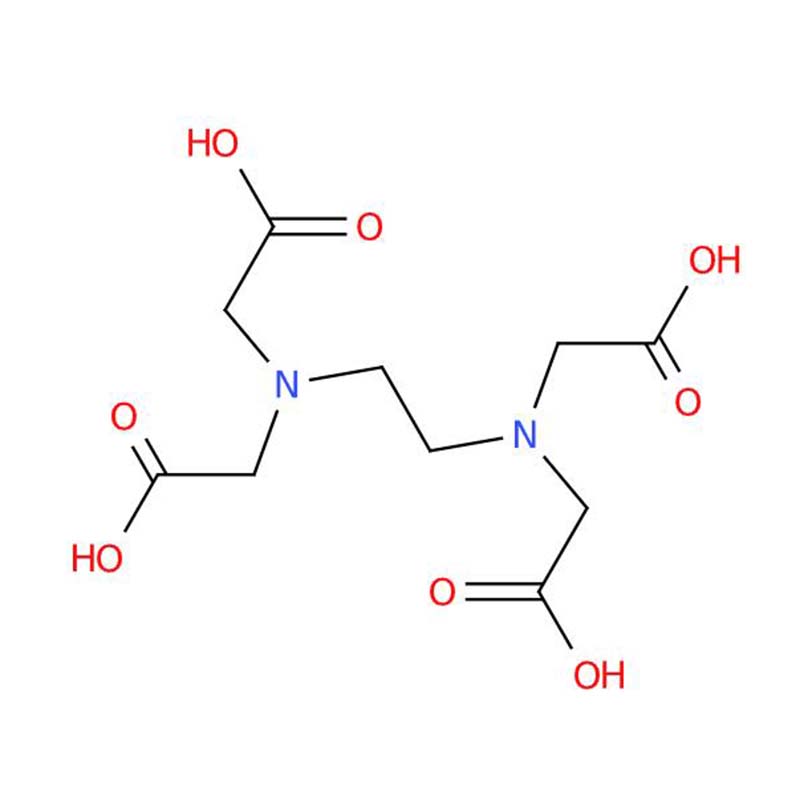
Ethylenediaminetetraacetic acid (EDTA) is a versatile aminopolycarboxylic acid and one of the most widely used chelating agents in the world. It is a colorless, water-soluble solid that forms stable complexes with metal ions (chelates), effectively binding and neutralizing them .
Its chemical formula is C₁₀H₁₆N₂O₈ with CAS number 60-00-4 and molecular weight of approximately 292.24 g/mol . The product typically appears as a white crystalline powder, practically odorless, with a slightly acidic taste.
EDTA is a hexadentate ligand, meaning it can form up to six coordinate bonds with a single metal ion. It was first described in 1935 by Ferdinand Münz, who synthesized it from ethylenediamine and chloroacetic acid. Today, it is produced globally in massive quantities and is essential across countless industrial, pharmaceutical, food, and household applications .
Its ability to sequester metal ions—preventing them from participating in unwanted reactions such as oxidation, precipitation, or microbial growth—makes EDTA an indispensable tool in modern chemistry and manufacturing .
2. Key Features
Exceptional Chelating Power: Forms stable, water-soluble complexes with most divalent and trivalent metal ions (Ca²⁺, Mg²⁺, Fe²⁺/³⁺, Cu²⁺, Zn²⁺, Pb²⁺, etc.). The stability constant for Fe³⁺-EDTA is particularly high (log K ≈ 25.1) .
Broad pH Applicability: Effective across a wide pH range, though chelation strength varies with pH. Maximum chelation typically occurs at alkaline pH where carboxyl groups are fully deprotonated .
Versatile Forms Available: Available in multiple salt forms (disodium EDTA, calcium disodium EDTA, tetrasodium EDTA) with different solubilities and applications, allowing formulation flexibility .
Highly Water Soluble (as salts) : While the free acid form has limited water solubility (~0.5 g/L at 25°C), its sodium salts are highly soluble (disodium EDTA: ~100 g/L; tetrasodium EDTA: ~600 g/L) .
Multi-Industry Essential: Indispensable across diverse sectors:
Pharmaceutical: Metal poisoning antidote, preservative, stabilizer
Food: Preservative (E385), color retention agent
Cosmetics: Stabilizer, preservative enhancer
Industrial: Water treatment, metal finishing, pulp/paper, textiles
Laboratory: Buffer component, metal ion masking agent
Agriculture: Micronutrient carrier
Biodegradable Alternatives Available: Responding to environmental concerns, readily biodegradable chelants (EDDS, MGDA, GLDA) are now available for applications where environmental persistence is a concern .
3. Technical Specifications with Explanations
| Parameter | Typical Value/Specification | Description & Significance |
|---|---|---|
| Molecular Formula | C₁₀H₁₆N₂O₈ | Core identifier for chemical structure; contains two amine and four carboxyl groups . |
| Molecular Weight | 292.24 g/mol | Used for quantitative analysis and formulation calculations . |
| CAS Number | 60-00-4 | Universal chemical substance identifier . |
| EINECS Number | 200-449-4 | European inventory identifier . |
| Appearance | White crystalline powder | Visual quality indicator; should be free from lumps, discoloration, or visible impurities . |
| Odor | Practically odorless | Sensory characteristic; no significant odor . |
| Assay (Purity) | ≥98.0% – ≥99.5% (grade dependent) | Core specification. Higher purity minimizes metal contamination and ensures consistent chelation capacity . |
| Melting Point | 237°C – 245°C (decomposes) | Characteristic physical constant; decomposition occurs upon heating rather than melting . |
| Solubility | Water (free acid): 0.5 g/L (25°C) NaOH solution: Soluble Disodium salt in water: ~100 g/L Tetrasodium salt in water: ~600 g/L Organic solvents: Insoluble | Critical formulation parameter. Free acid requires alkaline conditions to dissolve; salts provide water solubility . |
| pH (1% solution) | 2.5 – 3.5 (free acid suspension) | Free acid form is acidic; salts provide neutral to alkaline pH . |
| Heavy Metals (as Pb) | ≤ 10 – 20 ppm (grade dependent) | Critical safety parameter ensuring product purity for pharmaceutical and food applications . |
| Iron (Fe) | ≤ 10 – 50 ppm | Indicates contamination level; important for applications requiring metal-free conditions . |
| Sulfated Ash | ≤ 0.2% (free acid) | Measures inorganic impurities; low value indicates high purity . |
| Loss on Drying | ≤ 0.5% – 1.0% | Measures moisture content; critical for stability and accurate weighing . |
| Nitrilotriacetic Acid (NTA) | ≤ 0.1% | Controls potentially carcinogenic impurity; critical for pharmaceutical and food grades . |
| Chelation Capacity (mg CaCO₃/g) | ≥ 339 mg/g (EDTA acid basis) | Measures practical chelating power; 1g EDTA can chelate approximately 339 mg CaCO₃ equivalent . |
| pKa Values | pKa₁ = 0.26, pKa₂ = 0.96, pKa₃ = 2.67, pKa₄ = 6.16, pKa₅ = 10.26 | Acid dissociation constants determining chelation behavior at different pH levels . |
| Stability Constant (log K for Fe³⁺) | 25.1 | Indicates extremely strong binding with ferric iron; one of the highest among common metals . |
| Biodegradability | Poor (0-30% in 28 days) | Environmental persistence concern; biodegradable alternatives available . |
4. Applications
Pharmaceutical & Medical Applications
Heavy Metal Poisoning Antidote: Calcium disodium EDTA is FDA-approved for treating lead poisoning and other heavy metal toxicities (cadmium, mercury, zinc, manganese) . It works by binding the metal ions, allowing their excretion through urine .
Ophthalmic Preparations: Used as a preservative and stabilizer in eye drops to prevent metal-catalyzed degradation .
Topical Preparations: Prevents metal-induced oxidation in creams and ointments, extending shelf life .
Dental Applications: Root canal irrigants (e.g., 17% EDTA solution) to remove smear layer and clean dentin .
Anticoagulant: Added to blood collection tubes to prevent clotting by chelating calcium .
Pharmaceutical Excipient: Stabilizer in injectable formulations, preventing metal-catalyzed drug degradation .
Food & Beverage Industry (as E385)
Preservative: Prevents oxidation and discoloration in canned foods, seafood, dressings, and sauces by sequestering trace metals that catalyze rancidity .
Color Retention Agent: Maintains color in fruits, vegetables, and processed foods by preventing metal-induced color changes .
Antioxidant Synergist: Enhances effectiveness of antioxidants like BHA, BHT, and ascorbic acid .
Beverage Stabilizer: Precludes haze formation in soft drinks and beers by binding trace metals .
Typical Usage Levels: 25-800 ppm depending on application (regulated by food authorities) .
Cosmetics & Personal Care
Stabilizer: Prevents metal-catalyzed degradation of cosmetic ingredients, extending product shelf life .
Preservative Enhancer: Boosts effectiveness of traditional preservatives by chelating metals that bacteria need for growth .
Water Treatment: Improves performance of soaps and detergents by binding hard water ions (Ca²⁺, Mg²⁺) that cause soap scum .
Hair Care: Used in shampoos and conditioners to prevent metal-induced damage and improve lather .
Skin Care: Incorporated into creams and lotions to protect against metal-induced oxidation .
Industrial Applications
Water Treatment: Softens hard water by binding calcium and magnesium; controls scale formation in boilers and cooling towers .
Pulp & Paper Industry: Stabilizes hydrogen peroxide in bleaching processes by chelating metal ions that catalyze peroxide decomposition .
Textile Industry: Removes metal ions that interfere with dyeing processes; improves color brightness and uniformity .
Metal Finishing & Electroplating: Purifies plating baths by removing contaminating metals; improves deposit quality .
Chemical Cleaning: Component in industrial cleaners, boiler descaling formulations, and metal surface preparation .
Photography: Used in photographic developers to prevent precipitation of metal salts .
Rubber & Polymer Industry: Prevents metal-catalyzed degradation of polymers; stabilizes latex .
Laboratory & Research Applications
Buffer Component: Used in Tris-EDTA (TE) buffer for DNA/RNA storage and electrophoresis .
Enzyme Inhibition: Inhibits metalloproteases and other metal-dependent enzymes by removing essential metal cofactors .
Metal Ion Masking Agent: Prevents interference from metal ions in analytical procedures (titrations, colorimetric assays) .
Cell Culture: Added to media to prevent metal toxicity and maintain optimal ion concentrations .
Histology: Decalcification of bone and tissue samples for sectioning .
Agricultural Applications
Micronutrient Carrier: EDTA-chelated micronutrients (Fe-EDTA, Zn-EDTA, Mn-EDTA, Cu-EDTA) used in fertilizers to prevent precipitation and improve plant uptake .
Soil Remediation: Used in soil washing to remove heavy metal contaminants .
5. Product Grade Comparison
EDTA is available in multiple grades and salt forms to meet diverse application requirements:
| Parameter/Grade | Technical Grade | Industrial Grade | Food Grade (E385) | Pharmaceutical Grade (USP/Ph.Eur.) | ACS Reagent Grade | Analytical Standard |
|---|---|---|---|---|---|---|
| Core Standard | General industrial specs | Industrial applications | Meets food additive specifications | Meets USP/Ph.Eur. pharmacopoeia standards | Meets American Chemical Society specifications | ≥99.4% purity for precise analysis |
| Purity (Assay) | ≥98.0% | ≥99.0% | 99.0-100.5% (dried basis) | 98.0-102.0% (anhydrous basis) | 99.4-100.6% | ≥99.9% (HPLC) |
| Appearance | White powder | White powder | White powder | White powder | White powder | White powder |
| Heavy Metals (Pb) | ≤50 ppm | ≤20 ppm | ≤10 ppm | ≤10 ppm | ≤5 ppm | ≤5 ppm |
| Iron (Fe) | ≤100 ppm | ≤50 ppm | ≤50 ppm | ≤20 ppm | ≤5 ppm | ≤5 ppm |
| NTA Content | Not specified | Not specified | ≤0.1% | ≤0.1% | Not specified | Not specified |
| pH (1% solution) | 2.5-3.5 | 2.5-3.5 | 2.5-3.5 | 2.5-3.5 | 2.5-3.5 | 2.5-3.5 |
| Loss on Drying | ≤1.0% | ≤0.5% | ≤0.5% | ≤0.5% | ≤0.5% | ≤0.2% |
| Primary Applications | General chelation, water treatment, industrial cleaners | Metal finishing, textile processing, industrial manufacturing | Food preservation (E385), beverages, canned goods | Pharmaceuticals, ophthalmic preparations, parenteral formulations | Laboratory reagents, analytical chemistry, buffer preparation | HPLC calibration, quantitative analysis, reference standard |
| Packaging | 25 kg bags, 1000 kg supersacks | 25 kg bags, 200 kg drums | 25 kg drums, 5 kg containers | 1 kg, 5 kg, 25 kg with documentation | 100 g, 500 g, 1 kg, 5 kg bottles | 100 mg, 1 g, 5 g vials |
| Certifications | Manufacturer COA | Manufacturer COA | E385 compliance, KOSHER, Halal available | USP/Ph.Eur. certification, DMF available | ACS certificate | Traceable reference material |
Common Salt Forms Available:
| Salt Form | CAS Number | Water Solubility | Typical pH (1%) | Primary Applications |
|---|---|---|---|---|
| EDTA (Free Acid) | 60-00-4 | Low (0.5 g/L) | 2.5-3.5 | General chelation where acidity is acceptable |
| Disodium EDTA (Na₂EDTA) | 6381-92-6 (dihydrate) 139-33-3 (anhydrous) | ~100 g/L | 4.0-6.0 | Most common form for laboratory, pharmaceutical, and general use |
| Tetrasodium EDTA (Na₄EDTA) | 64-02-8 (anhydrous) 10378-23-1 (hydrate) | ~600 g/L | 10.5-11.5 | Industrial applications, alkaline detergents |
| Calcium Disodium EDTA | 62-33-9 (anhydrous) 23411-34-9 (hydrate) | ~200 g/L | 6.5-8.0 | Pharmaceutical (lead poisoning antidote), food preservation |
| Dipotassium EDTA | 2001-94-7 (anhydrous) 25102-12-9 (dihydrate) | High | 4.0-6.0 | Cosmetics, personal care |
6. Purchasing & Selection Guide
Select by Form & Grade
Pharmaceutical & Medical Applications:
Calcium Disodium EDTA USP: For lead poisoning treatment and chelation therapy .
Disodium EDTA USP/Ph.Eur.: For ophthalmic preparations, injectable formulations, and pharmaceutical excipient use .
Require Drug Master File (DMF) documentation and cGMP manufacturing .
Verify NTA content ≤0.1% and heavy metals ≤10 ppm .
Must meet sterility requirements for specific applications .
Food & Beverage Applications:
Select Food Grade (E385) disodium EDTA or calcium disodium EDTA .
Verify compliance with local regulations (FDA 21 CFR 172.135, EU Regulation 1333/2008) .
Request KOSHER/Halal certification if required .
Typical usage levels: 25-800 ppm depending on application .
Ensure documentation supports food additive status .
Laboratory & Analytical Applications:
ACS Reagent Grade (99.4-100.6%): For general lab use, buffer preparation, titrations .
Analytical Standard (≥99.9%): For HPLC calibration, quantitative analysis, reference standards .
Molecular Biology Grade: For DNA/RNA work (RNase/DNase-free) .
Cosmetics & Personal Care:
Disodium EDTA or Tetrasodium EDTA depending on formulation pH .
Focus on heavy metals control and microbial limits .
Verify compatibility with other ingredients .
Industrial & Technical Applications:
Acidic conditions: Free acid or disodium salt
Alkaline conditions: Tetrasodium salt
Technical/Industrial Grade: Cost-effective for water treatment, metal finishing, textiles .
Choose salt form based on solubility and pH requirements:
Important Selection Considerations
pH Requirements: Chelation efficiency varies with pH. Maximum chelation occurs at pH where carboxyl groups are deprotonated:
Ca²⁺, Mg²⁺: Optimal at pH 8-12
Fe³⁺: Effective even at low pH (2-3)
Zn²⁺, Cu²⁺: Optimal at pH 4-8
Metal Selectivity: EDTA binds metals in the following order of stability (highest to lowest):
Fe³⁺ > Cu²⁺ > Ni²⁺ > Pb²⁺ > Zn²⁺ > Co²⁺ > Fe²⁺ > Mn²⁺ > Ca²⁺ > Mg²⁺
Solubility Requirements:
Free acid requires NaOH or alkaline conditions to dissolve
Disodium salt is most versatile for general aqueous applications
Tetrasodium salt for high-concentration or alkaline formulations
Regulatory & Environmental Considerations
⚠️ Important Regulatory & Environmental Notes:
Biodegradability: EDTA is poorly biodegradable (0-30% in 28 days) and persists in the environment, raising concerns about metal mobilization in aquatic systems .
Alternatives: For environmentally sensitive applications, consider readily biodegradable chelants:
EDDS (Ethylenediamine-N,N'-disuccinic acid)
MGDA (Methylglycinediacetic acid)
GLDA (Glutamic acid diacetic acid)
IDS (Iminodisuccinic acid)
Food Use: Permitted in many countries with specific usage limits; verify local regulations .
Pharmaceutical Use: Must comply with relevant pharmacopoeia standards (USP, Ph.Eur., JP) .
REACH Status: Registered under EU REACH; compliance documentation available .
7. Frequently Asked Questions (FAQ)
Q: What is EDTA and how does it work?
A: EDTA (ethylenediaminetetraacetic acid) is a chelating agent that binds to metal ions, forming stable, water-soluble complexes. It works by surrounding the metal ion with six donor atoms (two nitrogen atoms from amines and four oxygen atoms from carboxyl groups), effectively "sequestering" the metal and preventing it from participating in unwanted reactions such as oxidation, precipitation, or catalysis .
Q: What is the difference between EDTA, disodium EDTA, and tetrasodium EDTA?
A: These are different forms of the same compound with varying degrees of neutralization:
EDTA (free acid): Fully protonated form; requires alkaline conditions to dissolve; lowest water solubility .
Disodium EDTA: Two carboxylic acid groups neutralized with sodium; most common laboratory form; soluble in water (~100 g/L); pH 4-6 .
Tetrasodium EDTA: Fully neutralized form; highly water soluble (~600 g/L); alkaline pH (10.5-11.5); preferred for industrial applications .
The choice depends on required solubility and formulation pH .
Q: Is EDTA safe for use in food and pharmaceuticals?
A: Yes, when used according to regulations. Calcium disodium EDTA and disodium EDTA are approved food additives (E385) and pharmaceutical excipients in many countries with specific usage limits . They have been extensively studied and are generally recognized as safe (GRAS) by FDA when used in compliance with good manufacturing practices . However, high-dose intravenous EDTA chelation therapy carries risks and should only be administered under medical supervision .
Q: How is EDTA used to treat heavy metal poisoning?
A: Calcium disodium EDTA is FDA-approved for treating lead poisoning and other heavy metal toxicities. The calcium in CaNa₂EDTA is displaced by toxic metals (lead, cadmium, mercury) with higher affinity for EDTA. The resulting metal-EDTA complex is water-soluble and excreted through urine, removing the toxic metal from the body . Treatment must be carefully monitored to avoid essential mineral depletion .
Q: Can EDTA be used as a preservative?
A: EDTA itself is not a traditional preservative but acts as a preservative enhancer by chelating metals that bacteria, yeast, and mold need for growth. It also prevents metal-catalyzed oxidation that degrades preservatives and other ingredients. In food, it preserves color, flavor, and texture by preventing metal-induced changes. In cosmetics, it boosts the effectiveness of traditional preservatives .
Q: Is EDTA biodegradable?
A: No, EDTA is poorly biodegradable (0-30% degradation in 28-day tests). It persists in the environment, leading to concerns about its potential to remobilize heavy metals from sediments and soils. For applications where environmental release is a concern, consider readily biodegradable alternatives such as EDDS, MGDA, or GLDA .
Q: How does pH affect EDTA chelation?
A: pH significantly impacts EDTA chelation because the carboxyl groups must be deprotonated to bind metals. At low pH, carboxyl groups are protonated and binding is weak. As pH increases, deprotonation occurs and chelation strengthens:
pH < 3: Fe³⁺ still binds strongly
pH 4-6: Good for Cu²⁺, Zn²⁺, Ni²⁺
pH 8-12: Optimal for Ca²⁺, Mg²⁺, and most divalent metals
The stability constants for each metal reflect binding strength at optimal pH .
Q: What is the stability constant of EDTA?
A: Stability constants (log K) vary by metal:
Fe³⁺: 25.1 (extremely stable)
Cu²⁺: 18.8
Ni²⁺: 18.6
Pb²⁺: 18.0
Zn²⁺: 16.5
Co²⁺: 16.3
Fe²⁺: 14.3
Mn²⁺: 13.9
Ca²⁺: 10.7
Mg²⁺: 8.7
Higher values indicate stronger binding. Fe³⁺-EDTA is one of the most stable metal complexes known .
Q: How should EDTA be stored?
A: Store in tightly sealed containers in a cool, dry, well-ventilated area away from strong oxidizing agents. Protect from moisture as EDTA is hygroscopic. Under recommended conditions, shelf life is typically 2-5 years depending on grade and packaging .
Q: What are the common uses of EDTA in the laboratory?
A: Common laboratory applications include:
TE buffer (Tris-EDTA) for DNA/RNA storage
Metal ion masking agent in analytical chemistry
Enzyme inhibition studies (metalloproteases)
Decalcification of tissue samples for histology
Titration for water hardness determination
Cell culture media to prevent metal toxicity
8. Delivery, Certification & Service
Delivery Capabilities
Inventory Status: Common grades and salt forms available from regional warehouses (US, Europe, Asia)
Sample Program: Small quantities available for research institutions and manufacturers for formulation testing and evaluation
Packaging Options:
Research quantities: 100g, 500g, 1kg, 5kg bottles or bags
Industrial quantities: 25kg bags or drums, 200kg drums, 1000kg supersacks
Bulk quantities: 20 MT containers for large-volume customers
Custom packaging: Available for volume customers (IBC totes, flexitanks)
Shipping: Non-hazardous material; standard freight shipping available (express, LTL, full truckload, container)
Quality Certifications
Certificate of Analysis (COA) provided with each batch for qualified grades, documenting:
Appearance
Assay/purity
Heavy metals (Pb, Fe, etc.)
Loss on drying
pH (for salt forms)
NTA content (where applicable)
Safety Data Sheet (SDS) available in multiple languages
Product Specification (PS) documenting chemical composition, physical properties, and storage requirements
Certificates of Origin (COO) available upon request for customs and regulatory compliance
Additional certifications available:
KOSHER certification
Halal certification
USP/Ph.Eur. certification for pharmaceutical grades
Food-grade (E385) compliance documentation
Drug Master File (DMF) for pharmaceutical applications
Technical Support & Services
Technical Consultation: Application specialist team available for formulation assistance, chelation guidance, and regulatory support
Custom Services: Tailored purity specifications, particle sizing, and packaging options for long-term partnership customers
Regulatory Support: Documentation packages for product registration in target markets, including food additive and pharmaceutical excipient dossiers
Documentation Available
Certificate of Analysis (COA)
Safety Data Sheet (SDS/MSDS)
Technical Data Sheet (TDS)
Product Specification (PS)
Certificate of Origin (COO)
KOSHER Certificate (where applicable)
Halal Certificate (where applicable)
USP/Ph.Eur. Certificate (pharmaceutical grades)
Food Grade Compliance Statement (E385)
GMP Declaration (pharmaceutical grades)
DMF Access Letter (upon qualification)