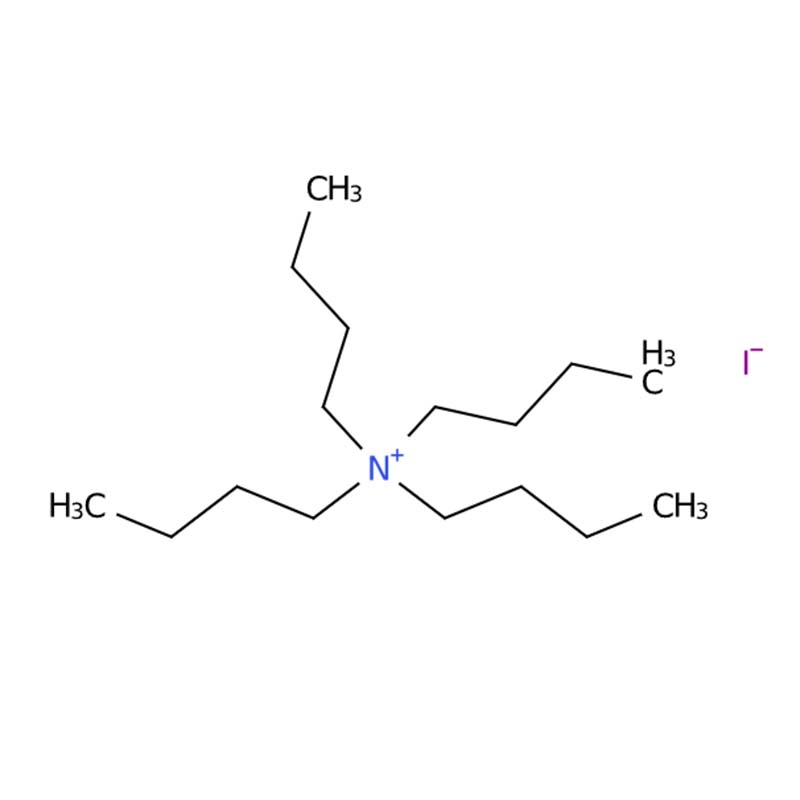
Tetrabutylammonium iodide
Highly Efficient Catalyst: Tetrabutylammonium iodide (TBAI) functions as an effective phase-transfer catalyst that significantly accelerates reaction rates.
Cost-Effective Iodine Source: It enables in-situ iodide generation, reducing dependence on unstable or expensive iodide reagents.
Versatile Application: The compound demonstrates broad utility across diverse chemical reactions, enhancing process flexibility.
Specialized Use in Dealkylation: Particularly effective for dealkylation reactions of esters and ethers, improving both efficiency and yield.
1. Product Overview
Tetrabutylammonium iodide (TBAI) is a quaternary ammonium salt widely recognized as one of the most effective phase-transfer catalysts in organic synthesis. Its chemical formula is (C₄H₉)₄N⁺ I⁻ or C₁₆H₃₆IN, with CAS number 311-28-4 and molecular weight of approximately 369.37 g/mol .
This compound features a bulky, lipophilic tetrabutylammonium cation paired with a simple iodide anion, creating a unique combination of properties that make it invaluable across multiple chemical disciplines. It appears as a white to off-white crystalline powder, stable under normal conditions but sensitive to light and moisture .
TBAI serves as a bridge between aqueous and organic phases, facilitating reactions that would otherwise proceed slowly or not at all due to immiscibility. Its ability to transfer ionic species across phase boundaries has made it indispensable in modern synthetic chemistry, from academic research laboratories to industrial-scale pharmaceutical manufacturing.
2. Key Features
Exceptional Phase-Transfer Catalysis: TBAI excels at transferring ionic reactants between immiscible phases (aqueous/organic), dramatically accelerating reaction rates and improving yields in biphasic systems . The bulky tetrabutylammonium cation effectively solubilizes anions in organic media .
Dual Functionality in Catalysis: Acts both as an iodide source and a phase-transfer facilitator in oxidative transformations. It enables in situ generation of reactive hypoiodite species for various functionalization reactions .
Broad Applicability Across Industries:
Organic Synthesis: Alkylation, acylation, ether cleavage, and nucleophilic substitutions
Pharmaceutical Research: Key intermediate in drug synthesis and API development
Materials Science: Defect passivation in perovskite solar cells
Electrochemistry: Supporting electrolyte and redox mediator
Polymer Science: Coinitiator in living cationic polymerization
High Purity Grades Available: Offered in multiple purity specifications from ≥98% to ≥99%, with Reagent Grade and higher purity options for sensitive applications .
Light Sensitivity Management: Available with light-protective packaging and storage recommendations to maintain stability and prevent decomposition .
Well-Characterized Safety Profile: Comprehensive hazard data available with established handling protocols for laboratory and industrial use .
3. Technical Specifications with Explanations
| Parameter | Typical Value/Specification | Description & Significance |
|---|---|---|
| Molecular Formula | C₁₆H₃₆IN or C₁₆H₃₆N·I | Quaternary ammonium salt structure with four butyl chains attached to nitrogen . |
| Molecular Weight | 369.37 - 369.38 g/mol | Used for stoichiometric calculations and solution preparation . |
| CAS Number | 311-28-4 | Universal chemical identifier . |
| EINECS Number | 206-220-5 | European inventory identifier . |
| MDL Number | MFCD00011636 | Available from multiple sources . |
| Appearance | White to almost white crystalline powder | Visual quality indicator; may appear slightly cream-colored . |
| Purity (Assay) | ≥98.0% - ≥99.0% (grade dependent) • ≥98.0% (argentometric/titration) • ≥99.0% (AT/titration) | Core specification. Higher purity critical for sensitive catalytic applications and pharmaceutical research. |
| Melting Point | 141°C - 149°C (varies by source) • 141-143°C • 143-146°C • 143-148°C • 144-149°C • 147°C | Characteristic physical constant for identification and purity assessment; narrow range indicates high purity. |
| Solubility | Water: Soluble Acetonitrile: 0.1 g/mL, clear, colorless Alcohol: Soluble Ether: Soluble Benzene: Insoluble | Critical formulation parameter. Solubility in both polar and non-polar solvents enables phase-transfer catalysis. |
| Density | 1.20 - 1.43 g/cm³ | Slightly denser than water; varies with measurement method . |
| Storage Temperature | 2-30°C (varies by grade) • 2-30°C • Below +30°C • 0-8°C • Cool place, <15°C recommended | Lower temperatures recommended for long-term stability, especially for high-purity grades. |
| Storage Conditions | Light Sensitive & Hygroscopic | Must be protected from light and moisture; store under inert gas recommended . |
| Shelf Life | Not specified; typically 2-5 years under proper conditions | Depends on storage conditions; protect from light and moisture. |
| Stability | Stable under recommended storage conditions; incompatible with strong oxidizing agents | Avoid contact with strong oxidizers; light-sensitive . |
| LD₅₀ (Oral, Rat) | 1,990 mg/kg | Acute toxicity value; harmful if swallowed . |
4. Applications
Organic Synthesis & Catalysis
Phase-Transfer Catalyst: TBAI facilitates reactions between organic and aqueous phases by transferring ionic species (e.g., nucleophiles, oxidizing agents) into organic media. This enhances reaction rates and selectivity in alkylation, acylation, and nucleophilic substitution reactions .
Oxidative α-Functionalization of Carbonyl Compounds: Catalyzes oxidative α-azidation of carbonyl compounds using sodium azide (NaN₃) and dibenzoyl peroxide (BPO). TBAI mediates in situ formation of ammonium hypoiodite species, promoting α-iodination followed by nucleophilic substitution via phase-transfer .
α-Oxyacylation of Ketones: Used with tert-butyl hydroperoxide (TBHP) for direct α-oxyacylation of ketones, providing efficient access to valuable synthetic intermediates .
Oxidative Coupling Reactions: Catalyzes metal-free, base-free oxidative coupling of enamides with sulfonylhydrazides to produce pharmaceutically relevant β-keto-sulfone compounds .
Hydroxylation of Naphthoquinones: Serves as an effective catalyst for hydroxylation of naphthoquinone derivatives using TBHP as oxidant, enabling synthesis of drug intermediates like parvaquone and lapachol .
Ether Synthesis and Cleavage: Used as a catalyst in the synthesis of ethers and in regioselective ether cleavage reactions .
Deprotection Reactions: Facilitates deprotection of benzyl (Bn) groups using phenyl trimethylsilyl sulfide and cleavage of methoxy groups with aluminum(III) chloride .
Materials Science & Optoelectronics
Perovskite Solar Cells (PSCs) : Acts as a surface passivation agent for all-inorganic CsPbI₂Br perovskite solar cells. TBAI treatment interacts with the Pb-I framework, passivating defect states and prolonging carrier lifetime, achieving champion power conversion efficiency (PCE) of 12.29% and retaining 90% of initial PCE after 300 hours in ambient air .
Polymer Blend Electrolytes: Optimizes polymer blend electrolytes in dye-sensitized solar cells (DSSCs). Addition of TBAI enhances ionic conductivity and overall performance, with specific weight ratios yielding highest efficiency .
Polymer Science
Living Cationic Polymerization: Acts as a coinitiator in living cationic polymerization of isobutyl vinyl ether. Concentration and nature of TBAI significantly influence polymerization rate and resulting polymer properties .
Electrochemistry
Supporting Electrolyte & Redox Mediator: Facilitates electrochemical synthesis of complex organic molecules, including phenanthridinones through constant-potential electrolysis. TBAI serves as both redox catalyst and supporting electrolyte .
Pharmaceutical & Medicinal Chemistry
Drug Intermediate Synthesis: Used in preparing bioactive compounds and pharmaceuticals, including hydroxylated naphthoquinones (parvaquone, lapachol) and β-keto-sulfone derivatives .
Quaternary Amine Antibacterial Agents: Employed in preparing novel quaternary amines to combat drug-resistant bacteria .
Analytical Chemistry
Chromatographic Reagent: Acts as a reagent in chromatographic techniques, aiding separation and analysis of complex mixtures .
Biotechnology
Biomolecule Extraction: Utilized for extraction and purification of biomolecules, enhancing yield and purity of desired products .
Corrosion Inhibition
Mild Steel Protection: Inhibits corrosion of mild steel in acidic environments through adsorption or complex formation on steel surface; effectiveness varies with concentration, temperature, and pH .
5. Product Grade Comparison
| Parameter/Grade | Synthesis Grade (≥98%) | Reagent Grade (≥99%) | High Purity (≥99.5%) | Analytical Standard |
|---|---|---|---|---|
| Core Standard | General organic synthesis | Meets reagent-grade specifications | Ultra-high purity for sensitive applications | ≥99.9% for precise analysis |
| Purity (Assay) | ≥98.0% (argentometric) | ≥99.0% (titration) | ≥99.5% | ≥99.9% (HPLC) |
| Melting Point | 143-146°C | 143-148°C | 144-149°C | Narrow, specified range |
| Storage Conditions | 2-30°C, protect from light | 0-8°C | Store under inert gas, cool & dark | Sealed, protected |
| Critical Parameters | Assay, melting point | Assay, melting point, appearance, solubility | Purity, heavy metals, iodide content, residual solvents | Exact content, chromatographic purity |
| Primary Applications | General phase-transfer catalysis, organic synthesis, academic research | Pharmaceutical research, sensitive catalytic reactions, electrochemistry | Perovskite solar cells, optoelectronic materials, drug synthesis, trace analysis | HPLC calibration, quantitative analysis, reference standard |
| Typical Packaging | 25g, 100g, 500g poly bottles | 25g, 100g, 500g poly bottles | 5g, 25g with inert atmosphere | 100mg, 1g vials |
6. Purchasing & Selection Guide
Select by Purity Grade
Synthesis Grade (≥98.0%) :
Suitable for general organic synthesis, academic research, and routine phase-transfer catalysis
Cost-effective for reactions where trace impurities do not affect outcomes
Typical applications: alkylation, acylation, ether synthesis, general laboratory use
Reagent Grade (≥99.0%) :
Required for pharmaceutical research, sensitive catalytic reactions, and electrochemistry
Higher purity ensures reproducibility in critical transformations
Typical applications: drug intermediate synthesis, oxidative functionalization, perovskite precursor preparation
High Purity (≥99.5%) :
Essential for optoelectronic materials (perovskite solar cells), trace analysis, and applications where impurities affect performance
Requires storage under inert gas with strict moisture/light protection
Typical applications: defect passivation in solar cells, electronic-grade materials
Select by Application-Specific Requirements
Phase-Transfer Catalysis: Standard purity (≥98%) sufficient for most transformations; focus on consistent quality and solubility characteristics .
Perovskite Solar Cells: Require highest purity (≥99.5%) with minimal metal impurities; must be stored under inert gas .
Pharmaceutical Intermediates: Reagent grade (≥99%) with documented purity profile and Certificate of Analysis .
Electrochemical Applications: High purity essential to avoid interfering redox processes .
Critical Selection Considerations
Light Sensitivity: All grades are light-sensitive; select packaging that provides light protection .
Hygroscopic Nature: TBAI absorbs moisture; choose appropriate packaging size to minimize exposure after opening .
Storage Temperature: Higher purity grades may require lower storage temperatures (0-8°C) for maximum stability .
Certificate of Analysis: Request COA to verify purity, melting point, and other specifications match application requirements .
7. Frequently Asked Questions (FAQ)
Q: What is Tetrabutylammonium Iodide (TBAI) and how does it work as a phase-transfer catalyst?
A: TBAI is a quaternary ammonium salt consisting of a large, lipophilic tetrabutylammonium cation and an iodide anion . As a phase-transfer catalyst, it facilitates reactions between immiscible phases (e.g., aqueous and organic) by transferring ionic species (reactants) into the organic phase where reactions occur. The bulky cation effectively solubilizes anions in organic media, dramatically accelerating reaction rates and improving yields in biphasic systems .
Q: What is the difference between TBAI and other tetrabutylammonium salts (e.g., TBAB, TBAF)?
A: The key difference lies in the anion. TBAI contains iodide (I⁻), while TBAB contains bromide (Br⁻), TBAF contains fluoride (F⁻), TBAH contains hydroxide (OH⁻), etc. TBAI is preferred for oxidative transformations because iodide can generate reactive hypoiodite species in situ . It also serves as an iodide source for nucleophilic displacement reactions . Choice depends on the specific reaction requirements.
Q: Is TBAI safe to handle? What are the main hazards?
A: TBAI is classified with the following hazards:
Harmful if swallowed (H302) — LD₅₀ oral rat: 1,990 mg/kg
Causes skin irritation (H315)
Causes serious eye irritation (H319)
May cause respiratory irritation (H335)
Signal word: Warning
Always use appropriate PPE (gloves, safety glasses), avoid dust formation, and work in well-ventilated areas .
Q: How should TBAI be stored to maintain stability?
A: TBAI is both light-sensitive and hygroscopic . Store in tightly sealed, light-protected containers in a cool, dry place. Recommendations vary by grade:
Standard grades: 2-30°C, protected from light
Reagent grade: 0-8°C recommended
High-purity grades: Store under inert gas
Avoid storage near strong oxidizing agents .
Q: What happens if TBAI is exposed to light or moisture?
A: Light exposure can cause decomposition, potentially leading to discoloration (yellowing/browning) and reduced catalytic activity. Moisture absorption can lead to caking and hydrolysis. Both effects degrade performance and purity. If discoloration occurs, the product's suitability should be verified before critical applications.
Q: What are the solubility characteristics of TBAI?
A: TBAI is soluble in water and many polar organic solvents, including acetonitrile (0.1 g/mL, clear, colorless) , alcohols, and ethers . It is insoluble in non-polar solvents like benzene . This unique solubility profile enables its function as a phase-transfer catalyst, bridging aqueous and organic phases.
Q: Why does TBAI have multiple melting point ranges reported?
A: Reported melting points range from 141-149°C depending on purity, measurement method, and crystalline form . Narrow ranges (e.g., 144-149°C) indicate higher purity. All values are within acceptable ranges for this compound.
Q: Can TBAI be used in pharmaceutical applications?
A: Yes, TBAI is widely used in pharmaceutical research and drug intermediate synthesis . However, for human or therapeutic use, appropriate grade selection (Reagent Grade or higher) with full quality documentation is essential. Standard TBAI is labeled "For R&D use only. Not for medicinal, household or other use" .
Q: How is TBAI synthesized?
A: TBAI is typically synthesized by reacting tetrabutylammonium hydroxide with hydroiodic acid . Alternatively, it can be prepared via anion exchange from tetrabutylammonium bromide with potassium iodide . Modern methods also include electrochemical synthesis and counterion exchange strategies .
Q: What is the role of TBAI in perovskite solar cells?
A: TBAI acts as a surface passivation agent for perovskite solar cells. When applied to CsPbI₂Br perovskite films, TBAI interacts with the Pb-I framework, passivating defect states and prolonging carrier lifetime. This improves power conversion efficiency and significantly enhances device stability (90% retention after 300 hours in ambient air) .
8. Delivery, Certification & Service
Delivery Capabilities
Inventory Status: Multiple grades available from regional warehouses
Sample Program: Small quantities available for research institutions and manufacturers for application testing
Packaging Options:
Research quantities: 25g, 100g, 500g poly bottles
Larger quantities: Available upon request
Light-protective packaging standard
Storage Recommendations: Transport and store at recommended temperatures; protect from light and moisture
Shipping: Non-hazardous under most transport regulations; consult SDS for specific requirements
Quality Certifications
Certificate of Analysis (COA) provided with each batch for qualified grades, documenting:
Appearance
Purity (titration/argentometric)
Melting point
Solubility characteristics
Safety Data Sheet (SDS) available in multiple languages
Product Specification (PS) documenting chemical composition and physical properties
Certificates of Origin (COO) available upon request
Additional certifications available:
Reagent Grade certification
High-purity documentation for specialized applications
Regulatory Compliance
EC Number: 206-220-5
RTECS#: BS5450000
HS Code: 2923900090
TSCA: Listed
WGK Germany: 2-3 (varies by source)
Technical Support & Services
Technical Consultation: Application specialists available for:
Phase-transfer catalysis optimization
Selection guidance for specific reactions
Storage and handling recommendations
Custom Services: Tailored packaging and purity specifications for volume customers
Regulatory Support: Documentation for research and development applications
Documentation Available
Certificate of Analysis (COA) with batch-specific data
Safety Data Sheet (SDS/MSDS)
Technical Data Sheet (TDS)
Product Specification (PS)
Certificate of Origin (COO)