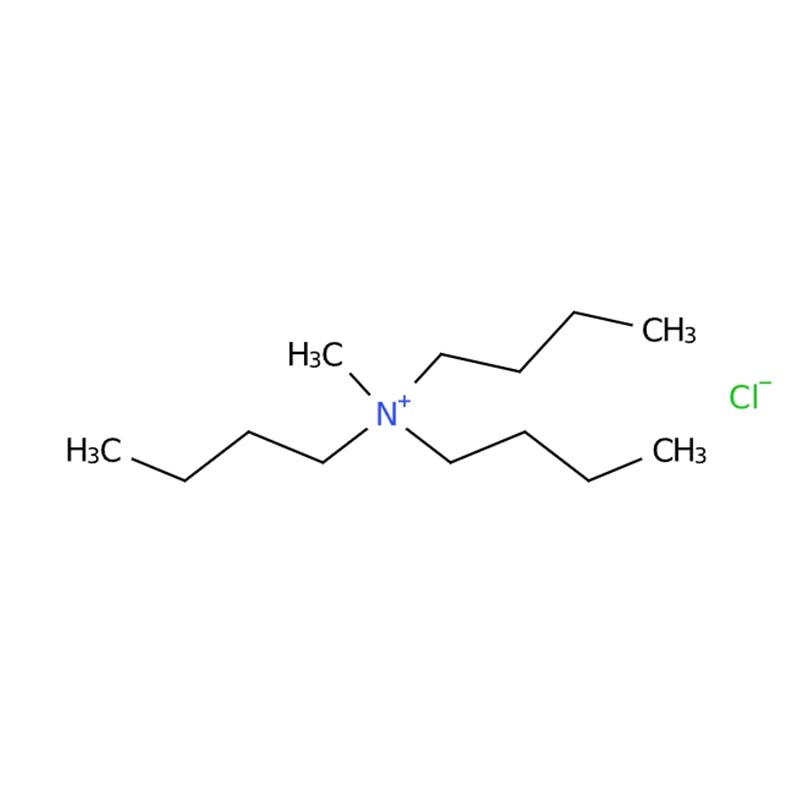
Methyl Tributyl Ammonium Chloride
Aqueous and Organic Solubility: Demonstrates ready solubility in aqueous, alcoholic, and ethereal solutions, enhancing formulation flexibility across multiple application fields.
Controlled Basicity: Exhibits mild alkaline characteristics under ambient conditions, providing advantages for specialized chemical synthesis and processing.
Thermal Endurance: Maintains structural integrity and functional performance under elevated temperature environments, ensuring operational reliability.
Chemical Resilience: Possesses robust molecular stability that guarantees consistent functionality throughout storage and application periods.
1. Product Overview
Methyl Tributyl Ammonium Chloride (MTBAC) is an important quaternary ammonium compound belonging to the asymmetric quaternary ammonium salt family. Its molecular structure features a positively charged nitrogen atom bonded to three butyl groups and one methyl group, with a chloride ion as the counterion. Its chemical formula is C₁₃H₃₀ClN with CAS number 56375-79-2 and molecular weight of approximately 235.84 g/mol .
MTBAC is recognized industrially as one of the most cost-effective phase-transfer catalysts available. Compared to symmetrical tetrabutylammonium bromide (TBAB), it offers significant cost advantages while providing excellent catalytic performance . Its unique asymmetric structure makes the charge center more accessible, resulting in superior reaction kinetics in phase-transfer catalysis applications .
The product is available in multiple forms, most commonly as a 75% aqueous solution (e.g., Aliquat® 175) or as ≥98% crystalline solid . It is widely used in organic synthesis,
pharmaceuticals, agrochemicals, and materials science, playing a critical role in facilitating heterogeneous reactions .
2. Key Features
Highly Efficient Phase-Transfer Catalyst (PTC) : Effectively transfers ionic reactants from aqueous or solid phases into organic phases, dramatically accelerating reaction rates and improving yields in biphasic systems .
Significant Cost Advantage: Production costs are lower than traditional symmetrical quaternary ammonium catalysts (e.g., tetrabutylammonium bromide/chloride), offering a more economical choice while maintaining catalytic efficiency .
Unique "Asymmetric" Structural Advantage: Unlike symmetrical TBAB, MTBAC's methyl and tributyl structure creates a charge center with reduced steric hindrance and enhanced accessibility, improving catalytic activity and selectivity in certain reactions .
Excellent Solubility and Easy Separation: Good water solubility allows easy removal of the catalyst from the organic phase by simple water washing after reaction completion, simplifying workup procedures .
Multiple Product Forms Available: Available in both 75% aqueous solution (easy to meter and add, safe to handle, low viscosity) and 98-99% crystalline solid (for anhydrous or moisture-sensitive reaction systems) to meet diverse process requirements .
Broad Applicability: Serves not only as a phase-transfer catalyst but also as a metal extractant, catalyst in antioxidant synthesis, and surfactant in specialized formulations .
3. Technical Specifications with Explanations
| Parameter | Typical Value/Specification | Description & Significance |
|---|---|---|
| Molecular Formula | C₁₃H₃₀ClN | Core identifier for chemical structure . |
| Molecular Weight | 235.84 g/mol | Used for stoichiometric calculations and solution preparation . |
| CAS Number | 56375-79-2 | Universal chemical substance identifier . |
| EC Number | 260-135-8 | European inventory identifier . |
| Appearance | Colorless to pale yellow transparent liquid (75% solution) White to off-white crystalline solid (98-99%) | Visual quality indicator; different forms correspond to different grades . |
| Purity (Assay) | 75 wt. % in H₂O (aqueous solution) ≥98% / ≥99% (crystalline solid) | Core specification. Solution form for direct use; high-purity solid for anhydrous or precision synthesis . |
| Melting Point | 95 - 99°C (crystalline solid) 105 - 108°C (98% solid) -33°C (aqueous solution) | Characteristic physical constant for solid products; solution remains liquid at low temperatures . |
| Boiling Point | 84-85°C (at 101 mmHg) | Distillation data at specified pressure . |
| Density | ~0.964 g/cm³ (aqueous solution) | Used for volume-to-weight conversions . |
| Refractive Index | n20/D 1.439 (aqueous solution) | Optical property for quality control . |
| Solubility | Soluble in water and most organic solvents | Good water solubility is key for phase-transfer catalysis and easy separation . |
| Sensitivity | Hygroscopic (solid form) | Solid form requires sealed storage to prevent moisture absorption . |
| Flash Point | Not applicable (aqueous solution) / 84-85°C (organic solutions) | Aqueous solution has no flash point; organic solutions require fire safety precautions . |
4. Applications
Organic Synthesis & Catalysis
Phase-Transfer Catalysis (Core Application) : MTBAC catalyzes reactions by transferring active anions from inorganic salts (KOH, NaOH, KCN, NaCN) or oxidizing agents into the organic phase :
Nucleophilic Substitution: Halide displacement, ester synthesis
C-Alkylation & O-Alkylation: O-alkylation of phenols, C-alkylation of active methylene compounds
Baeyer-Villiger Oxidation: Catalyzes oxidation of cyclohexanone to ε-caprolactone (polymer monomer)
Dichlorocarbene Reactions: Generates dichlorocarbene under strong alkaline conditions for addition to alkenes
Pharmaceutical & Fine Chemical Industry
Pharmaceutical Intermediate Synthesis: Enables difficult reactions in complex drug molecule construction, improving selectivity and yield of target products
Chiral Synthesis: Serves as a base structure for developing asymmetric phase-transfer catalysts
Metal Extraction & Separation
Extractant: Used in hydrometallurgy as an extractant for metal complexes, separating and purifying specific metal ions
Polymer Science
Polymer-Supported Catalysts: Can be immobilized on polymer supports to create insoluble triphase catalysts, facilitating catalyst recovery and continuous production processes
Other Industrial Applications
Surfactant: Functions as a surfactant or emulsifier in specialized formulations due to its quaternary ammonium structure
Antioxidant Additives: Acts as a catalyst in the synthesis of antioxidants
5. Product Grade Comparison
| Parameter/Grade | 75% Aqueous Solution | 98-99% Crystalline Solid | High Purity (≥99.5%) |
|---|---|---|---|
| Typical Form | Colorless to pale yellow liquid, low viscosity | White to off-white crystalline solid or paste | White crystalline solid |
| Typical Purity | 75 wt% | ≥98% / ≥99% | ≥99.5% |
| Representative Trade Names | Aliquat® 175 | Generic MTBAC | High-purity MTBAC |
| Melting Point | ~ -33°C (solution) | 95 - 108°C (solid) | 105-108°C |
| Handling Convenience | Excellent. Easy to pump, meter, and add; no melting required | Requires melting or dissolution in solvents; more complex handling | Requires careful handling under inert atmosphere |
| Suitable Reaction Systems | Conventional aqueous-organic biphasic systems | Anhydrous or moisture-controlled reaction systems | Ultra-sensitive systems, trace analysis |
| Storage & Transport | Protect from freezing; winter insulation may be required | Highly hygroscopic; strict sealing and dry storage essential | Sealed under inert gas, desiccated |
| Primary Applications | Routine phase-transfer catalysis, large-scale production, extraction | Anhydrous reactions, precision synthesis, catalyst immobilization | Pharmaceutical reference standards, advanced research |
6. Purchasing & Selection Guide
Select by Reaction System
Conventional PTC Reactions with Aqueous Phase:
Choose 75% Aqueous Solution. It is ready to use, requires no heating, and is ideal for industrial production .
Anhydrous or Strongly Basic Reactions:
Must select 98-99% Crystalline Solid. Water from the solution form could interfere with the reaction or cause side reactions.
Select by Purity Requirements
For most catalytic reactions, trace impurities in the catalyst typically do not affect results, so 98% purity is sufficient .
For pharmaceutical excipient synthesis or precision electrochemical studies, ≥99% or ≥99.5% high-purity grade is recommended.
Consider Cost-Effectiveness
Compare the cost-performance ratio of TBAB (tetrabutylammonium bromide) and MTBAC. MTBAC typically offers lower molar cost, making it a smart choice for cost reduction without compromising catalytic efficiency .
Storage Considerations
Solid MTBAC is highly hygroscopic. Verify packaging integrity (e.g., foil bags, fluorinated bottles). Use promptly after opening and store remaining material in a desiccator with strict sealing .
Aqueous solutions require freeze protection in winter (store above freezing point).
7. Frequently Asked Questions (FAQ)
Q: What is the difference between Methyl Tributyl Ammonium Chloride (MTBAC) and Tetrabutylammonium Bromide (TBAB)?
A: Key differences include:
Structure: MTBAC is asymmetric (three butyl, one methyl); TBAB is symmetric (four butyl).
Anion: MTBAC contains chloride; TBAB contains bromide.
Cost & Activity: MTBAC offers lower cost and, due to its asymmetric structure, may show higher catalytic activity in certain reactions. TBAB, while widely used, typically carries a higher cost .
Q: How should solid Methyl Tributyl Ammonium Chloride be stored?
A: Solid MTBAC is highly hygroscopic and must be stored in tightly sealed containers in a cool, dry, well-ventilated area away from direct moisture. After removing product, immediately reseal the container tightly. Storage in a desiccator is recommended for opened containers .
Q: My 75% aqueous solution froze during winter shipping. Is it ruined?
A: No. The 75% aqueous solution has a low freezing point (approximately -33°C) . If frozen under extreme conditions, simply thaw slowly at room temperature and mix or shake thoroughly before use. The product quality remains unaffected by this reversible physical change.
Q: Is MTBAC toxic? What precautions should I take?
A: MTBAC is classified as harmful if swallowed (H302) and causes skin and eye irritation (H315, H319) . Always use appropriate personal protective equipment (lab coat, safety goggles, chemical-resistant gloves). Work in a well-ventilated area and avoid inhaling dust or vapors.
Q: In which classic organic reactions is MTBAC particularly effective?
A: MTBAC excels in dichlorocarbene generation, cyanidation reactions, esterification, alkylation reactions (particularly O-alkylation and C-alkylation), and Baeyer-Villiger oxidations .
8. Delivery, Certification & Service
Delivery Capabilities
Inventory Status: Common sizes of 75% aqueous solution and 98% crystalline solid available from regional warehouses .
Sample Program: Small quantities available for research institutions and manufacturers for catalyst screening and process validation.
Packaging Options:
Research quantities: 100g, 500g fluorinated bottles or foil bags (solid); 500mL, 1L plastic bottles (liquid)
Industrial quantities: 25 kg plastic drums (liquid) or fiber drums with polyethylene liner (solid)
Bulk quantities: IBC totes (liquid) or 500 kg supersacks (solid)
Shipping: Not classified as hazardous under most regulations . Ensure packaging integrity, especially for solid products.
Quality Certifications
ISO 9001:2015 certified manufacturing facilities
Certificate of Analysis (COA) provided with each batch, documenting:
Appearance
Purity (titration/GC)
Water content (solid grades)
Other critical parameters
Safety Data Sheet (SDS) available in multiple languages
Third-party test reports (e.g., SGS) available upon request
Technical Support & Services
Technical Consultation: Chemist team available for online consultation on phase-transfer catalysis applications, catalyst selection, and process optimization
Custom Services: Tailored concentration specifications (e.g., 50% solutions) and packaging for qualified long-term customers
Regulatory Support: Documentation for export and product registration
Documentation Available
Certificate of Analysis (COA)
Safety Data Sheet (SDS/MSDS)
Technical Data Sheet (TDS)
Product Specification (PS)
Certificate of Origin (COO)