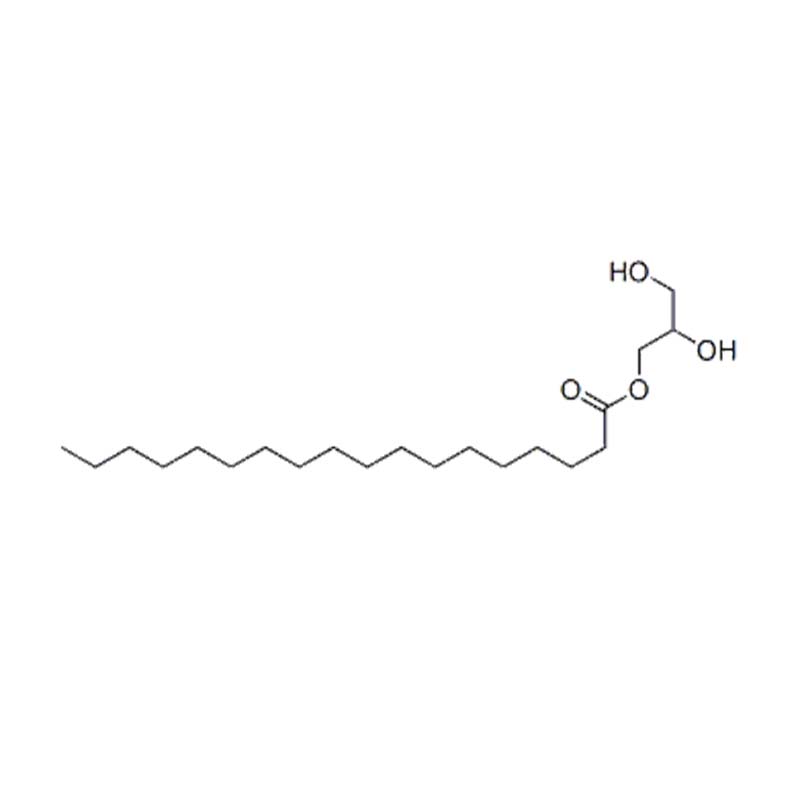
Glyceryl monostearate
Emulsifying Performance: With an HLB value of 3.8, it demonstrates effective emulsifying capabilities, making it particularly useful in industrial applications that require stable water-in-oil emulsions.
Versatile Solubility Profile: It dissolves readily in hot organic solvents and oils, but remains insoluble in water, allowing flexible use across diverse formulation systems.
Safety for Use in Food: Recognized as safe by the FAO/WHO with no specified limit on Acceptable Daily Intake (ADI), supporting its use in food-grade and industrial products.
Thermal Stability: Exhibits a high boiling point of 410.96°C and a melting point between 78–81°C, reflecting its stability under high-temperature processing conditions.
1. Product Overview
Glyceryl Monostearate (GMS) is a lipophilic non-ionic surfactant belonging to the glycerol mono-fatty acid ester family. It is produced through the esterification of glycerol with stearic acid (derived from vegetable or animal fats and oils) or through glycerolysis of fats and oils, resulting in a mixture of mono-, di-, and triglycerides .
Its chemical formula is typically represented as C₂₁H₄₂O₄ with molecular weight of 358.6 g/mol and CAS number 31566-31-1 . Commercial GMS appears as white to cream-colored waxy flakes, beads, powders, or pellets, solid at room temperature .
Glyceryl monostearate is one of the oldest and most widely used emulsifiers globally. Its molecular structure features both hydrophilic hydroxyl groups (-OH) and a lipophilic long alkyl chain, enabling it to significantly reduce interfacial tension between oil and water phases, forming stable emulsions . It is an indispensable ingredient across the food, pharmaceutical, cosmetic, and industrial sectors.
2. Key Features
Excellent Emulsification & Stabilization: Functions as an effective water-in-oil (W/O) or oil-in-water (O/W) emulsifier, forming stable emulsions and preventing phase separation. Acts as a thickener and stabilizer, imparting smooth texture to products like ice cream and creams .
Multifunctional Versatility: Serves not only as an emulsifier but also as a:
Pharmaceutical excipient: Sustained-release matrix material, tablet lubricant
Cosmetic ingredient: Emollient, plasticizer, pigment dispersant
Food additive: Starch complexing agent, texture modifier
High Safety Profile: Recognized as Generally Recognized as Safe (GRAS) by the FDA for food use . Generally regarded as non-toxic and non-irritating in cosmetic and pharmaceutical applications .
Excellent Compatibility: Compatible with other co-emulsifiers, oils, and active ingredients. When combined with PEG-75 stearate, it can stabilize challenging active pharmaceutical ingredient (API) systems .
Controllable Quality & Sourcing: Available in various purity grades (40-55% monoester standard grade to ≥90% high-purity grade) and sources (vegetable-derived or synthetic) to meet specific application requirements .
Good Thermal Stability: High melting point (≥55°C) ensures stability during heating, emulsification, and cooling processes. GMS-based oleogels demonstrate excellent shelf-life and thermal stability for sustained drug delivery applications .
3. Technical Specifications with Explanations
Based on USP/NF, Ph. Eur., JP, and food additive standards:
| Parameter | Typical Value/Specification | Description & Significance |
|---|---|---|
| CAS Number | 31566-31-1 (primary) 11099-07-3 | Universal chemical identifiers; different CAS numbers may correspond to different grades or sources . |
| Molecular Formula | C₂₁H₄₂O₄ | Idealized formula for glyceryl monostearate . |
| Molecular Weight | 358.6 g/mol | Used for stoichiometric calculations and formulation . |
| HLB Value | 3.0 – 4.0 (pure monoester) 8 ± 1 (blends with PEG stearate) | Core functional parameter. HLB 3-4 indicates strong lipophilic character for W/O emulsions; blends provide higher HLB for O/W systems . |
| Appearance | White to cream-colored waxy flakes, beads, powder, or pellets | Visual quality indicator; different physical forms suit different processing needs . |
| Melting Point | ≥55°C 55.5 – 62.5°C | Core physical parameter. Solid below this temperature, liquid above; critical for structuring and thermal processing . |
| Monoester Content | 40-55% (standard grade) ≥90% (high purity grade) | Key purity indicator. Higher monoester content indicates higher purity and emulsifying efficiency. Commercial GMS contains varying proportions of di- and triesters . |
| Acid Value | Increases with storage time | Measures free fatty acid content. Low acid value indicates freshness and stability; improper storage (heat exposure) increases acid value . |
| Iodine Value | Depends on raw material source | Measures unsaturation. GMS produced from hydrogenated stearic acid has very low iodine value, indicating excellent oxidative stability . |
| Density | 1.0 ± 0.1 g/cm³ | Slightly denser than water; used for volume-to-weight conversions . |
| Solubility | Insoluble in water, dispersible in hot water; soluble in hot organic solvents (ethanol, oils) | Determines processing requirements—must be heated to dissolve in oil phase and disperse in water . |
| Log P (Partition Coefficient) | 7.23 | Extremely high lipophilicity; strongly partitions into oil phase . |
| Storage Conditions | Store in sealed containers in cool, dry place; protect from light | Prevents moisture absorption, oxidation, and acid value increase . |
4. Applications
Food Industry
Baked Goods: Functions as a dough conditioner and anti-staling agent in bread and cakes, improving texture and retarding starch retrogradation to maintain freshness .
Dairy & Frozen Desserts: Acts as an emulsifier and stabilizer in ice cream, controlling fat agglomeration to produce smooth, creamy texture with good melt resistance. Stabilizes emulsions in whipped toppings and margarine .
Confectionery: Reduces viscosity and prevents fat bloom in chocolate; serves as an emulsifier in toffees and caramels to prevent oil separation .
Other Applications: Stabilizer in protein beverages; thickener and color stabilizer in flavors and fragrances .
Pharmaceutical Industry
Pharmaceutical Excipient: Functions as a tablet lubricant and coating agent, facilitating compression during tablet manufacturing .
Sustained-Release Formulations: Serves as a matrix-forming agent for oral sustained-release tablets, pellets, and veterinary boluses, controlling drug release kinetics .
Long-Acting Injectable Systems: Recent research demonstrates GMS-based oleogels as effective long-acting local anesthetic delivery systems, providing significantly prolonged anesthesia duration compared to liposomal bupivacaine through sustained drug release .
Emulsifying Agent: Used in preparing O/W or W/O emulsion bases for creams, ointments, and suppositories .
Cosmetics & Personal Care
Skin Care & Color Cosmetics: Widely used as an emulsifier, emollient, and thickener in creams, lotions, foundations, and sunscreens, providing stable texture and silky feel .
Hair Care: Functions as an emulsifier and conditioning agent in conditioners and hair masks, imparting smoothness and manageability .
Other Applications: Used in antiperspirants, baby care products, and other personal care formulations .
Industrial Applications
Plastics Industry: Acts as a lubricant and antistatic agent in resin processing .
Textile Industry: Component in fiber finishing oils and processing aids .
5. Product Grade Comparison
| Parameter/Grade | Standard Grade (40-55%) | High Purity Grade (≥90%) | Self-Emulsifying Grade | Pharmaceutical Grade (USP/NF) |
|---|---|---|---|---|
| Core Standard | Meets food additive standards (FCC, E471) | High monoester content for specific applications | Contains added emulsifiers (e.g., soaps, PEG esters) for self-emulsification | Meets USP/NF or Ph.Eur. pharmacopoeia standards |
| Monoester Content | 40-55% (with di- and triesters) | ≥90% | Variable (typically 30-50% with co-emulsifiers) | Meets monograph requirements |
| HLB Value | 3-4 | 3-4 | 8 ± 1 (higher due to co-emulsifiers) | 3-4 (pure) or blended |
| Physical Form | Flakes, beads, powder | Flakes, powder, pellets | Flakes, beads | Flakes, powder |
| Critical Parameters | Acid value, melting point, monoester content | Monoester content, acid value, heavy metals | Self-emulsifying properties, HLB | Heavy metals, microbial limits, purity, GMP compliance |
| Primary Applications | Bakery, dairy, confectionery, general food applications | High-quality cosmetics, pharmaceutical excipients, sustained-release formulations | Cosmetics requiring simple O/W emulsification (creams, lotions) | Oral tablets, topical creams/ointments, sustained-release systems |
| Typical Packaging | 25 kg bags/drums | 20 kg, 25 kg bags/drums | 25 kg drums | 1 kg, 5 kg, 25 kg with documentation |
6. Purchasing & Selection Guide
Select by Application Area
Food Manufacturers:
Standard Grade (40-55% monoester) suitable for most food applications (bakery, dairy, confectionery)
Verify compliance with local regulations (FDA 21 CFR, EU E471)
Request Certificate of Analysis confirming acid value, melting point, and heavy metals compliance
Pharmaceutical Manufacturers:
Certificate of Analysis with batch-specific data
Drug Master File (DMF) access
cGMP manufacturing certification
Must select Pharmaceutical Grade meeting USP/NF or Ph.Eur. standards
Require comprehensive documentation:
For sustained-release formulations, verify monoester content and purity
For topical applications, ensure appropriate grade selection
Cosmetic Formulators:
High Purity Grade (≥90% monoester) for premium formulations requiring maximum purity and performance
Self-Emulsifying Grade for simplified O/W emulsion preparation
Verify cosmetic-grade specifications including color, odor, and heavy metals
Research Applications:
High Purity Grade for sustained-release research, oleogel studies
Request Certificate of Analysis with complete characterization
Critical Selection Considerations
Monoester Content: Higher monoester content = higher purity and emulsifying efficiency
Physical Form: Flakes for general use; powder for easier dissolution; pellets for handling
Source: Verify vegetable/animal source based on application requirements (vegan, kosher, halal)
Self-Emulsifying Grades: Contain added emulsifiers; verify HLB value for your specific application
Regulatory Compliance: Ensure grade meets target market regulations for intended application
7. Frequently Asked Questions (FAQ)
Q: What is the HLB value of glyceryl monostearate and what does it mean?
A: Pure glyceryl monostearate has an HLB value of 3.0-4.0 , indicating strong lipophilic character suitable for water-in-oil (W/O) emulsions and as a stabilizer in oil-in-water (O/W) emulsions when combined with high-HLB emulsifiers. Self-emulsifying grades containing co-emulsifiers have higher HLB values (e.g., 8 ± 1 ) for simplified O/W emulsion preparation .
Q: What is the difference between standard GMS and high-purity GMS?
A: Standard GMS contains 40-55% monoester along with di- and triglycerides, making it suitable for general food and industrial applications. High-purity GMS contains ≥90% monoester, offering higher emulsifying efficiency and purity for demanding pharmaceutical, cosmetic, and research applications .
Q: Is glyceryl monostearate safe for use in food and cosmetics?
A: Yes, GMS has an excellent safety profile. It is GRAS (Generally Recognized as Safe) by the FDA for food use and is widely accepted as non-toxic and non-irritating in cosmetic and pharmaceutical applications . It is approved as food additive E471 in the EU.
Q: How should glyceryl monostearate be stored?
A: Store in tightly sealed containers in a cool, dry, well-ventilated area away from direct sunlight, heat sources, and strong oxidizing agents . Protect from moisture. Under proper conditions, shelf life is typically 2 years . Improper storage can lead to increased acid value and degradation .
Q: How do I incorporate GMS into my formulation?
A: Due to its high melting point (≥55°C), GMS must be incorporated by heating. For emulsions:
Add to oil phase ingredients
Heat together to 65-75°C (above melting point) until completely melted and dissolved
Combine with heated water phase with mixing
For self-emulsifying grades, follow manufacturer's specific instructions
Q: Can GMS be used in vegan products?
A: Yes, but depends on the stearic acid source. Vegetable-derived GMS (from palm or coconut oil) is suitable for vegan formulations. Always verify the source with your supplier and request documentation for vegan certification if required .
Q: What is the difference between GMS and self-emulsifying GMS?
A: Self-emulsifying GMS contains added emulsifiers (such as soaps, PEG esters, or other surfactants) that allow it to form O/W emulsions more easily, often without the need for additional emulsifiers. It has a higher HLB value (typically 8 ± 1) compared to pure GMS (HLB 3-4) .
Q: What is the role of GMS in sustained-release pharmaceutical formulations?
A: GMS serves as a hydrophobic matrix-forming agent in oral sustained-release tablets and pellets. When combined with drugs, it forms a matrix that slowly erodes or allows drug diffusion over extended periods, controlling release kinetics . Recent research also demonstrates its utility in long-acting injectable oleogels .
8. Delivery, Certification & Service
Delivery Capabilities
Inventory Status: Multiple grades available from regional warehouses (Asia, Europe, Americas)
Sample Program: Small quantities available for qualified customers for formulation testing and evaluation
Packaging Options:
Research quantities: 1 kg, 5 kg bags/containers
Industrial quantities: 25 kg multi-layer paper bags, fiber drums, or cartons
Bulk quantities: 500 kg supersacks, palletized, 20 MT containers
Custom packaging: Available for volume customers
Shipping: Non-hazardous material; standard freight shipping available
Quality Certifications
Certificate of Analysis (COA) provided with each batch for qualified grades, documenting:
Appearance and color
Melting point
Acid value
Monoester content
Saponification value (where applicable)
Iodine value (where applicable)
Heavy metals (where applicable)
Safety Data Sheet (SDS) available in multiple languages
Product Specification (PS) documenting chemical and physical properties
Certificates of Origin (COO) available upon request
Additional certifications available:
USP/NF/Ph.Eur. certification for pharmaceutical grades
Food-grade (E471) compliance documentation
KOSHER certification
HALAL certification
ISO 9001 quality management certification
GMP documentation for pharmaceutical grades
Non-GMO declaration
Allergen statement
Regulatory Compliance
Food Grade: Meets FCC, E471 specifications
Pharmaceutical Grade: Listed in USP/NF and Ph. Eur.
FDA: Recognized as GRAS for food use
Export Documentation: Full compliance package for international shipping
Technical Support & Services
Technical Consultation: Application specialists available for:
Grade selection guidance
Formulation development support
Emulsification optimization
Sustained-release formulation development
Regulatory compliance questions
Custom Services: Tailored packaging, blending, and quality specifications for qualified customers
Regulatory Support: Documentation for global compliance and product registration
Documentation Available
Certificate of Analysis (COA) with batch-specific data
Safety Data Sheet (SDS/MSDS)
Technical Data Sheet (TDS)
Product Specification (PS)
Certificate of Origin (COO)
KOSHER certificate
HALAL certificate
Non-GMO declaration
Allergen statement
ISO certifications
GMP compliance statement (pharmaceutical grades)
USP/Ph.Eur. certification