Methyl Red
Precise pH Indicator: Methyl Red serves as a highly reliable acid-base indicator with a well-defined transition range of pH 4.4-6.2, enabling clear differentiation between acidic (red) and basic (yellow) conditions during titrations.
Sharp Endpoint Detection: The compound's vivid color shift at the equivalence point enables precise determination of titration endpoints in analytical procedures.
Organic Solubility Profile: While demonstrating limited aqueous solubility, it readily dissolves in ethanol, chloroform, and acetic acid, providing formulation flexibility in various solvent systems.
Stable Material Characteristics: Supplied as either lustrous purple crystals or reddish-brown powder, the reagent maintains consistent physical and chemical properties under standard laboratory conditions.
1. Product Overview
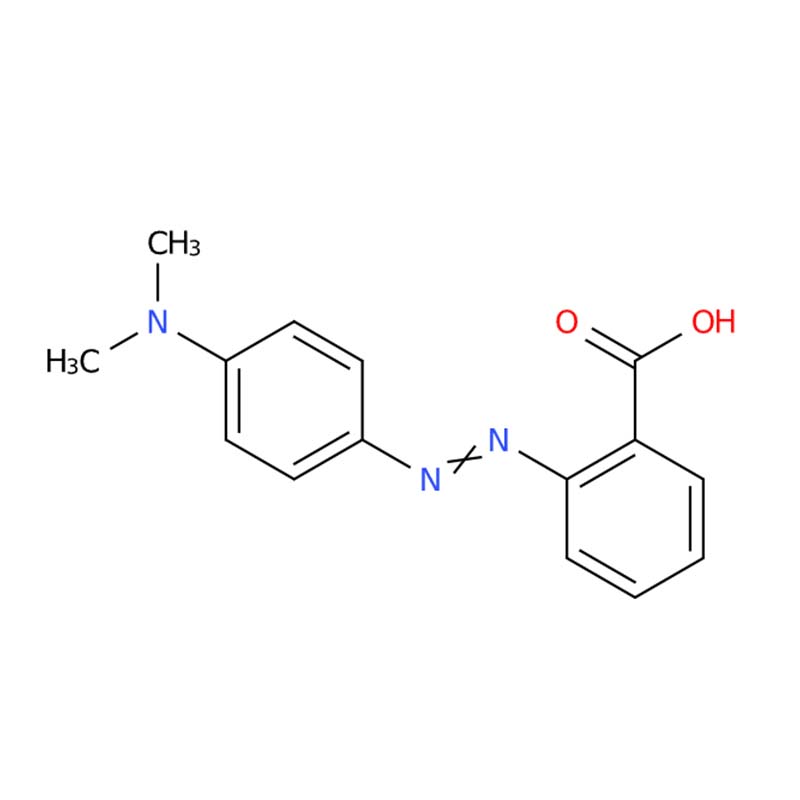
Methyl Red (also known as C.I. Acid Red 2, 2-[(4-Dimethylamino)phenylazo]benzoic acid) is a synthetic azo dye widely recognized as one of the most reliable pH indicators in analytical chemistry . With the chemical formula C₁₅H₁₅N₃O₂ and CAS number 493-52-7, this compound exhibits a distinctive and sharp color transition from red to yellow across the acidic pH range .
As an aromatic azo compound belonging to the azobenzene class, Methyl Red features a characteristic azo group (-N=N-) conjugated to benzene rings, which is responsible for its chromophoric properties . Beyond its classic role as a pH indicator, Methyl Red serves critical functions in microbiological diagnostics, clinical biochemistry, and various analytical determination methods .
The product typically appears as lustrous violet crystals or reddish-brown powder, with excellent stability when properly stored, though its ethanolic solutions may gradually esterify over extended periods .
2. Key Features
Sharp, Distinct Color Transition — Exhibits a vivid and easily distinguishable color change from red (pH <4.2) to yellow (pH >6.2) , with an orange intermediate zone between pH 4.4–6.2 . This clear visual endpoint makes it ideal for titration applications.
Dual-Functionality — Serves both as a pH indicator and as a biological stain. In the Methyl Red (MR) Test, it is the essential reagent for differentiating enteric bacteria based on glucose fermentation patterns .
Versatile Analytical Applications — Functions effectively in multiple analytical techniques:
Acid-base titrations (ammonia, weak organic bases, alkaloids)
Adsorption indicator in precipitation titrations (fluoride titration with thorium nitrate)
Oxidizing agent detection (free chlorine, chlorites)
Mixed Indicator Compatibility — Can be combined with bromocresol green or methylene blue to create mixed indicators with narrower transition ranges and enhanced endpoint sharpness .
Multiple Grade Options — Available in various purity grades (IND, pure, >97%, EP Grade) to suit different application requirements from routine teaching labs to regulated pharmaceutical testing .
Ready-to-Use Solutions — Available as pre-prepared solutions (e.g., 50 mg/100 mL) manufactured to European Pharmacopoeia standards, eliminating preparation time and ensuring batch-to-batch consistency .
3. Technical Specifications with Explanations
| Parameter | Typical Value | Description & Significance |
|---|---|---|
| Molecular Formula | C₁₅H₁₅N₃O₂ | Azo dye structure with carboxylic acid and dimethylamino functional groups responsible for pH-dependent color change . |
| Molecular Weight | 269.3 g/mol | Used for stoichiometric calculations and solution preparation . |
| CAS Number | 493-52-7 | Primary chemical registry identifier . |
| Color Index No. | 13020 | Dye classification number for industrial and textile applications . |
| Appearance | Lustrous violet crystals or reddish-brown powder | Visual quality indicator; color intensity relates to purity and crystal form . |
| Melting Point | 178°C – 182°C | Characteristic physical property; sharp melting range indicates high purity . Narrow range confirms product quality. |
| pH Transition Range | Red: pH <4.2–4.5 Orange: pH 4.4–6.2 Yellow: pH >6.2 | Critical functional parameter. The exact transition point varies slightly by source (4.2–6.2 or 4.4–6.2) but always provides a clear visual endpoint . |
| Transition Intervals | - 4.2 (red) to 6.2 (yellow) - 4.4 (red) to 6.2 (yellow) - 4.5 (red) to 6.2 (yellow) (Chinese HG standard) | Minor variations exist between different standards and manufacturers; all provide reliable acid-base indication. |
| Absorption Maxima (λmax) | pH 4.5: 523–528 nm pH 6.2: 427–437 nm | Spectral characteristics for spectrophotometric applications; the bathochromic shift confirms the color transition mechanism . |
| Mass Absorption Coefficient | α₁ (pH 4.5): ≥130 L/cm·g α₂ (pH 6.2): ≥70 L/cm·g | Quantitative measure of color intensity; higher values indicate stronger absorbance and better visual sensitivity . |
| Solubility | Almost insoluble in water; soluble in ethanol and acetic acid | Critical for proper solution preparation. Must be dissolved in alcohol or alcohol/water mixtures for aqueous applications . |
| Drying Loss | ≤1.0% | Maximum allowable moisture/content volatility; ensures accurate weight-based preparation . |
| Ignition Residue | ≤0.2% (as sulfate) | Measure of inorganic impurity content; important for applications requiring high purity . |
| Ethanol Solubility Test | Passes (complete solution) | Confirms absence of insoluble impurities that could affect solution preparation . |
4. Applications
Analytical Chemistry & Titration
Acid-Base Indicators: Classical indicator for titrations involving strong acids with weak bases, ammonia, and certain alkaloids
Mixed Indicators: Combined with bromocresol green or methylene blue for enhanced endpoint sharpness in complexometric and precipitation titrations
Adsorption Indicator: Used in precipitation titrations (e.g., fluoride determination with thorium nitrate) where color change occurs on precipitate surfaces
Clinical & Microbiological Diagnostics
Methyl Red (MR) Test: Essential reagent in the IMViC panel for identifying enteric bacteria (e.g., E. coli, Enterobacter). Bacteria producing stable acids from glucose fermentation turn the medium red .
Clinical Serum Protein Testing: Used in biochemical assays for protein determination and analysis
Oxidizing Agent Detection
Free Chlorine/Chlorite Detection: Color change indicates presence of oxidizing agents in water and solution testing
Biosensor Development
Glucose Biosensors: Functions as a pH indicator in enzymatic glucose detection systems, particularly for saliva analysis applications
Carbohydrate and Lactic Acid Detection: Used in various enzymatic assay configurations
Research Applications
Protozoan Vital Staining: Used for staining living protozoan specimens for microscopic observation
Environmental Microbiology: Studies on bacterial decolorization of azo dyes for bioremediation research
5. Product Grade Comparison
| Parameter/Grade | Indicator Grade (IND) | Pure Grade | EP Grade Solution | Research Grade (>97%) |
|---|---|---|---|---|
| Purity | Meets HG/T 3449—2012 standard | High purity, suitable for general lab use | Exact concentration: 50 mg/100 mL | >97% |
| Form | Powder/crystals | Powder/crystals | Ready-to-use liquid | Powder/crystals |
| Packaging | 25 g, 100 g, 500 g glass bottles | 25 g, 100 g, 500 g glass bottles | 100 mL tamper-evident bottles | Various |
| Quality Specifications | pH transition: 4.5–6.2 λmax verified Mass absorption: ≥130/70 Drying loss ≤1.0% Ignition residue ≤0.2% | Melting point: 178–182°C Infrared spectrum authenticated | Manufactured to Eu. Ph. Chapter 4 (4.1.1) Certificate of Analysis provided Expiration dated | Minimum purity >97% for general research |
| Primary Applications | Analytical titration Pharmacopoeia compliance Mixed indicators | General laboratory Teaching labs Routine analysis | Clinical microbiology (MR Test) Pharmaceutical quality control Regulated environments | Research applications Biosensor development Exploratory studies |
| Shelf Life/Stability | Stable when sealed dry; ethanolic solutions may esterify over time | Store in cool, dry place away from oxidizers | Expiration dated; ready-to-use stability | Store protected from light and moisture |
| Safety Classification | Warning; harmful if swallowed | Warning; harmful if swallowed | Danger; flammable liquid, eye irritant | Warning; harmful if swallowed |
6. Purchasing & Selection Guide
Select by Application
Indicator Grade (IND) — Recommended for analytical laboratories requiring compliance with national standards (HG/T 3449—2012). Suitable for pharmacopoeia methods, quality control, and educational institutions performing precise titrations.
Pure Grade — Ideal for general laboratory use, teaching laboratories, and routine analytical work where pharmacopoeia compliance is not required. Cost-effective for high-volume applications.
EP Grade Solution — Essential for clinical microbiology performing the Methyl Red (MR) Test for bacterial differentiation. The ready-to-use format eliminates preparation variability and ensures compliance with European Pharmacopoeia standards.
Research Grade (>97%) — Suitable for biosensor development, enzymatic assays, and exploratory research where exact purity requirements may vary.
Formulation Considerations
Powder Form: Offers maximum stability and flexibility; requires dissolution in ethanol before use. Preferred for laboratories preparing their own reagents.
Solution Form: Eliminates preparation time and variability; ensures consistent concentration; ideal for clinical labs with high throughput.
Stability & Storage Notes
Store in tightly sealed containers in cool, dry conditions away from strong oxidizing agents
Ethanolic solutions may gradually esterify over extended storage, reducing indicator sensitivity—prepare fresh solutions periodically
Protect from prolonged light exposure to prevent photodegradation of the azo bond
7. Frequently Asked Questions (FAQ)
Q: What is the exact pH range of Methyl Red as an indicator?
A: Methyl Red typically transitions from red at pH ≤4.2–4.5 to yellow at pH ≥6.2, with orange shades between pH 4.4–6.2 . Minor variations exist between different national standards, but all provide reliable endpoints for acid-base titrations.
Q: How do I prepare Methyl Red indicator solution?
A: Dissolve the powder in ethanol first, as Methyl Red is almost insoluble in water. A common preparation: dissolve 0.1 g in 100 mL of 95% ethanol. For aqueous use, dilute the ethanolic solution with water as needed .
Q: What is the Methyl Red (MR) test in microbiology?
A: The MR test identifies enteric bacteria that perform mixed acid fermentation of glucose. Bacteria producing sufficient stable acids lower the medium pH below 4.4, causing the added Methyl Red indicator to turn red (positive result). Bacteria not producing these acids result in a yellow color (negative result) .
Q: Why does my Methyl Red solution lose sensitivity over time?
A: Ethanol solutions of Methyl Red can undergo slow esterification of the carboxylic acid group during prolonged storage, which alters the indicator's properties . Prepare fresh solutions periodically (e.g., monthly) for optimal performance.
Q: Can Methyl Red be used for titrating organic acids?
A: Methyl Red is suitable for titrating ammonia, weak organic bases, and alkaloids. However, it is not recommended for titrating organic acids except for oxalic acid and picric acid .
Q: Is Methyl Red safe to handle?
A: Methyl Red is classified as harmful if swallowed (Warning). Solutions may be flammable (especially ethanol-based preparations) and can cause eye irritation. Always use appropriate personal protective equipment (gloves, safety glasses) and work in a well-ventilated area .
8. Delivery, Certification & Service
Delivery Capabilities
Inventory Status: Common sizes (25 g, 100 g) available from regional warehouses
Sample Program: Small quantities available for evaluation
Bulk Quantities: 500 g packaging for high-volume laboratories
Solution Formats: 100 mL ready-to-use solutions with tamper-evident packaging
Hazardous Material Shipping: Compliant with international dangerous goods regulations for ethanol-based solutions
Quality Certifications
Certificate of Analysis (COA) provided with each production lot for EP Grade and upon request for other grades
ISO 9001 certified manufacturing facilities for select grades
European Pharmacopoeia (Eu. Ph.) compliance for EP Grade solutions (Chapter 4, 4.1.1)
Chinese National Standard (HG/T 3449—2012) compliance for Indicator Grade
Merck Index referenced (14,6119) for research applications
Technical Support
Application Specialists: Available for assistance with titration methods, MR test protocols, and analytical procedures
Method Development Support: Guidance on mixed indicator preparation and optimization
Custom Formulations: Custom concentrations and packaging available for volume customers
Documentation Available
Safety Data Sheets (SDS) in multiple languages
Certificate of Analysis with batch-specific test results
Regulatory compliance documentation for pharmaceutical and clinical laboratories